��Ŀ����
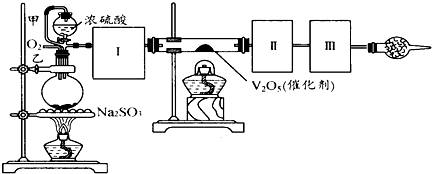
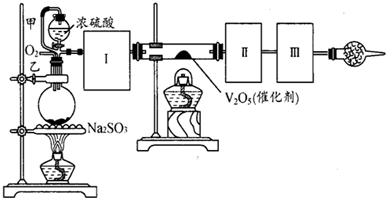
����ͼװ�ÿ��Խ��вⶨSO2ת����SO3��ת���ʵ�ʵ�顣��֪SO3���۵��� 16��8�棬�е���44��8�档��֪����װ�������漰��Ӧ�Ļ�ѧ����ʽΪ��
Na2SO3��s����H2SO4��80���� Na2SO4��SO2����H2O
��ע��80��H2SO4����Ũ��������ԡ�
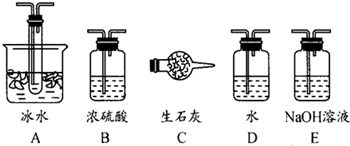
��1������ʵ����Ҫ��Ӧ���ڢ����Ӻ��ʵ�װ�á������ͼA��Dװ����ѡ������ʵ�װ�ò����������������Ŀո��С�
�����ӵ�װ�÷ֱ���___________��_____________��_____________��
��2����ʵ���С����ȴ������롰�μ�Ũ���ᡱ�IJ���������Ӧ��ȡ�IJ�����___________��
��3����һС����ʵ���з��֣�SO2����������������º���ʵ���������ԣ����ֲ��������������⣬�����Ʋ���ܵ�ԭ��˵����Ӧ����֤������
��ԭ��__________________________________________________��
��֤����________________________________________________��
��ԭ��__________________________________________________��
��֤����________________________________________________��
��4��������SO2ͨ�뺬1��0mol���������Һ�У���1��204��1024������ת�ƣ���������ǿ�ᣬ�÷�Ӧ�Ļ�ѧ����ʽΪ______________________________��
��5����25��2gNa2SO3��ĩ������Ũ������д�ʵ�飬����Ӧ����ʱ������ͨ��O2һ��ʱ�佫��Ӧ������SO2ȫ���ų������װ�â�������3��2g����SO2��ת����Ϊ__________��
����:��

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д� Na2SO4+SO2��+H2O��ע��80%H2SO4����Ũ��������ԣ�
Na2SO4+SO2��+H2O��ע��80%H2SO4����Ũ��������ԣ�