��Ŀ����
����Ũ�Ⱦ�Ϊ0.1mol/L��������Һ�������ᡢ���������ơ����Ȼ�李��ܴ����ƣ���ش��������⣺��1��25��ʱ���١��ڡ��ۡ���������ҺpH�ɴ�С��˳���ǣ�����ţ�______��
��2��25�棬���ںܵ͢������Ϻ��Һ�и�����Ũ���ɴ�С��˳����______��
��3����֪t�棬KW=1×10-13����t�棨�����������=����______25�森��t��ʱ����pH=11��NaOH��Һa L��pH=1��H2SO4����Һb L��ϣ����Ի�Ϻ���Һ����ı仯���������û����Һ��pH=2����a��b=______��
���𰸡���������1������Խǿ��pHԽС������Һ��pH��ˮ���Լ��Ե�����Һ��pH��ˮ�������Ե�����Һ��pH������Һ��pH��
��2���ںܵ͢������ϣ���Һ�Լ��ԣ���Һ�д��ڵ�����NaOH��NaAc��
��3�������¶ȣ��ٽ�ˮ�ĵ��룬KwԽ���¶�Խ����t��ʱ����pH=11��NaOH��Һa L��pH=1��H2SO4����Һb L��ϣ����Ի�Ϻ���Һ����ı仯���������û����Һ��pH=2�����������������ʣ�������ӵ�Ũ�ȼ���pH��
����⣺��1������Խǿ��pHԽС����ͬŨ��ʱ�ɼ���Һ��pH��ˮ���Լ��Ե�����Һ��pH��ˮ�������Ե�����Һ��pH������Һ��pH��
��١��ڡ��ۡ���������ҺpH�ɴ�С��˳��Ϊ�ڣ��ܣ��ۣ��٣��ʴ�Ϊ���ڣ��ܣ��ۣ��٣�
��2���ںܵ͢������ϣ���Һ�Լ��ԣ���Һ�д��ڵ�����NaOH��NaAc�����������ˮ���Լ��ԣ�
��c��OH-����c��CH3COO-���������������ӣ���������Ũ�����
���Ի��Һ�и�����Ũ���ɴ�С��˳��Ϊc��Na+����c��OH-����c��CH3COO-����c��H+����
�ʴ�Ϊ��c��Na+����c��OH-����c��CH3COO-����c��H+����
��3��t�棬KW=1×10-13��1×10-14���ٽ���ˮ�ĵ��룬��t�棾25�棬
t��ʱ����pH=11��NaOH��Һa L��pH=1��H2SO4����Һb L��ϣ����Ի�Ϻ���Һ����ı仯����
�����û����Һ��pH=2��
��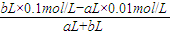 =0.01mol/L�����a��b=9��2��
=0.01mol/L�����a��b=9��2��
�ʴ�Ϊ������9��2��
���������⿼��pH���йؼ��㣬��ϤKw�������ӵ�Ũ�ȵĻ����ǽ��Ĺؼ���ע�⣨3�����¶Ȳ���25��Ϊ�����״��㣬��Ŀ�Ѷ��еȣ�
��2���ںܵ͢������ϣ���Һ�Լ��ԣ���Һ�д��ڵ�����NaOH��NaAc��
��3�������¶ȣ��ٽ�ˮ�ĵ��룬KwԽ���¶�Խ����t��ʱ����pH=11��NaOH��Һa L��pH=1��H2SO4����Һb L��ϣ����Ի�Ϻ���Һ����ı仯���������û����Һ��pH=2�����������������ʣ�������ӵ�Ũ�ȼ���pH��
����⣺��1������Խǿ��pHԽС����ͬŨ��ʱ�ɼ���Һ��pH��ˮ���Լ��Ե�����Һ��pH��ˮ�������Ե�����Һ��pH������Һ��pH��
��١��ڡ��ۡ���������ҺpH�ɴ�С��˳��Ϊ�ڣ��ܣ��ۣ��٣��ʴ�Ϊ���ڣ��ܣ��ۣ��٣�
��2���ںܵ͢������ϣ���Һ�Լ��ԣ���Һ�д��ڵ�����NaOH��NaAc�����������ˮ���Լ��ԣ�
��c��OH-����c��CH3COO-���������������ӣ���������Ũ�����
���Ի��Һ�и�����Ũ���ɴ�С��˳��Ϊc��Na+����c��OH-����c��CH3COO-����c��H+����
�ʴ�Ϊ��c��Na+����c��OH-����c��CH3COO-����c��H+����
��3��t�棬KW=1×10-13��1×10-14���ٽ���ˮ�ĵ��룬��t�棾25�棬
t��ʱ����pH=11��NaOH��Һa L��pH=1��H2SO4����Һb L��ϣ����Ի�Ϻ���Һ����ı仯����
�����û����Һ��pH=2��
��
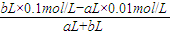 =0.01mol/L�����a��b=9��2��
=0.01mol/L�����a��b=9��2���ʴ�Ϊ������9��2��
���������⿼��pH���йؼ��㣬��ϤKw�������ӵ�Ũ�ȵĻ����ǽ��Ĺؼ���ע�⣨3�����¶Ȳ���25��Ϊ�����״��㣬��Ŀ�Ѷ��еȣ�

��ϰ��ϵ�д�
 ��У��������ĩ��̾�ϵ�д�
��У��������ĩ��̾�ϵ�д�
�����Ŀ