��Ŀ����
�ҹ�����ר�Һ�°����ڴ��£��Ľ��������ˡ������Ƽ����Ϊ�����Ƽҵ������ͻ�����ס�������������⣺
��1���������Ƽ���Ƶõġ���� (�ѧʽ)��
��2������������Ƽ��������Ҫ�Ĺ�ҵ�Ƽ�����б����У�����ȷ���� ��
| | | ��� | �����Ƽ |
| A | ԭ�� | ʳ�Ρ���������ʯ�� | ʳ�Ρ�������������̼ |
| B | ���ܵĸ����� | �Ȼ��� | �Ȼ�� |
| C | ѭ������ | ������������̼ | �Ȼ��� |
| D | ���� | ԭ���ã��豸���ӣ��ܺĸ� | ԭ�������ʸߣ��������� |
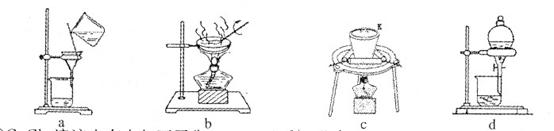
ijʵ��С�飬��������װ��ģ�⡰�����Ƽ����

��3��ȡ������������װ�ã�˳��Ϊ��(a)��( )��( )��( )��(b)��( )��
���������Ժ�װ��ҩƷ��Ӧ������ װ�ã���������ĸ���ȷ�����Ӧ��ֱ�����������岻������C���ܽ�ʱ����ͨ����һװ���в��������塣
��4��C�������θ���ܶ�����ֱ���ܣ��������� ��D��Ӧѡ�õ�Һ��Ϊ ��
��5��C�й��ƿ�ڲ���������ܻ�ѧ����ʽΪ ��
��6����Ʒ�����к���̼�����ơ�����ü��ȷֽ�ķ����ⶨ������̼�����Ƶ�����������������̼�����Ƶ����������ɱ�ʾΪ�� (ע����ı���ʽ�����õ��йط��ŵĺ���)��
��1��Na2CO3
��2��A.C
��3��f e d c ��2�֣� B
��4�������� ����NaHCO3��Һ
��5��CO2+NH3+NaCl+H2O��NaHCO3��+NH4Cl
��6�� �� m1Ϊ��Ʒ������m2Ϊ���Ⱥ��Ʒ��������
�� m1Ϊ��Ʒ������m2Ϊ���Ⱥ��Ʒ��������
���������������1���������Ƽ���Ƶõġ����̼���ƣ���ѧʽΪNa2CO3��
��2��A�����ԭ���У�ʳ�Σ��Ȼ��ƣ���ʯ��ʯ��������������ʯ�ҺͶ�����̼���������������Ƽԭ���У�ʳ�Ρ�������������̼����A����B��������ܵĸ�����Ϊ�Ȼ��ƣ������Ƽ���ܵĸ������Ȼ�泥���B��ȷ��C�����ѭ�����ʣ�������������̼�������Ƽѭ�����ʣ��Ȼ��ƣ�������̼����C����D�����ԭ�ϣ�ʳ�κ�ʯ��ʯ�����ˣ���Ʒ����Ĵ��ȸߣ�����Ʒ���Ͷ�����̼�����Ի���ѭ��ʹ�ã����첽����ʺ��ڴ��ģ���������豸���ӣ��ܺĸߣ���������ȱ�㻹����ԭ��ʳ�ε�������ֻ��72%��74%�������Ƽ�����ŵ���ʹʳ�ε���������ߵ�96%���ϣ��������٣���D��ȷ����ѡAC��
��3��Aװ�����Ʊ�CO2��Bװ�����Ʊ�NH3�����ڰ�����������ˮ��Ҫ�ø���ܷ�ֹ����������CO2�е��Ȼ������廹��Ҫ��ȥ�������ȷ������˳��Ϊ(a)��(f)��(e)��(d)��(b)��(c)������CO2��ˮ�е��ܽ��С�����Ҫ���Ȳ���������Ȼ����ͨ��CO2���壬����Ӧ������Bװ���ȷ�����Ӧ��
��4��������������ˮ�����C�������θ���ܶ�����ֱ���ܵ������Ƿ���������ȥCO2�е��Ȼ�������Ӧ���ñ���NaHCO3��Һ����D��Ӧѡ�õ�Һ��Ϊ����NaHCO3��Һ��
��5��Cװ�����Ʊ�̼�����Ƶģ�����C�й��ƿ�ڲ���������ܻ�ѧ����ʽΪCO2+NH3+NaCl+H2O��NaHCO3��+NH4Cl��
��6����m1Ϊ��Ʒ������m2Ϊ���Ⱥ��Ʒ�������������̼�����Ʒֽ�ķ���ʽ��֪
2NaHCO3 Na2CO3��H2O��CO2�� �����������١�m
Na2CO3��H2O��CO2�� �����������١�m
168g 106g 62g
X m1��m2
���X��
���Դ�����̼�����Ƶ����������ɱ�ʾΪ ��
��
���㣺��������Ƽ���й��жϡ������

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д���1���֣�
һλͬѧ�ڸ�ϰʱ��������һ��ϰ�⣺ij��ɫ��Һ�п��ܺ��С�H+��OH-��Na+��NO3-�����������ۺ�ֻ����H2���ʸ���ɫ��Һ���ܴ��������ļ������ӡ�
��1���������۲���H2��˵��������______��������ԡ���ԭ�ԡ�����
��2����ͬѧ��������H+�������ڣ���NO3-�Ͳ��ܴ������ڡ�
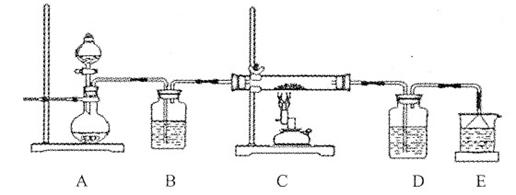
���ʵ��֤ʵ���£�
| װ �� | �� �� |
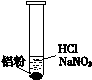 | ��. ʵ���ʼ��δ���������� ��. ��һ������������ݣ�Һ���Ϸ���dz��ɫ ��. �Թܱ��ȣ���Һ���� |
�� �����ܽ�Al2O3��Ĥ�����ӷ���ʽ��______��
�� ���������Ʋ���Һ�в�����NO��Ϊ��һ��ȷ�ϣ���������ʵ�飺
| ʵ �� | �� �� | �� �� |
| ʵ��1 | ��ʪ��KI��������ֽ���ڿ����� | δ���� |
| ʵ��2 | ��ʪ��KI��������ֽ����dz��ɫ���� | ��ֽ���� |
b. ʵ��1��Ŀ����_______��
c. ʵ��1��2˵����Ӧ������NO��������NO�����ӷ���ʽ����������
��3���ټ��裺��OH-�������ڣ�NO3-Ҳ���ܲ��ܴ������ڡ�
�������ʵ��֤ʵ���£�
| װ �� | �� �� |
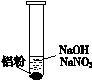 | ��. ʵ���ʼ��δ���������� ��. ��һ������������ݣ��д̼�����ζ |
Ϊȷ�ϡ��̼�����ζ�����壬��������ʵ�飺��ʪ��KI��������ֽ���飬δ��������ʪ���ɫʯ����ֽ���飬��ֽ������
�� �̼�����ζ��������______��
�� ��������������ӷ���ʽ��______��
��4����NaOH��Һ�м������ۣ����ֻ�������H2���ɣ��仯ѧ����ʽ��______��
��5��ʵ����֤ʵ��NO3?���ᡢ���Ի����ж���һ���������ԣ������������ʣ��������������ϰ���е���ɫ��Һһ���ܴ������ڵ���Na+��OH-��
ʵ��Ŀ�ģ�̽������������ˮ��Ӧ�����Һ�μӷ�̪��Һ�ȱ�����ɫ��ԭ��
[���������]
��1�����ݹ���������ˮ��Ӧ��ԭ����2Na2O2 + 2H2O =" 4NaOH" + O2�������������ƹ�����ȫ�ܽⷴӦ�����Һ�еμӷ�̪��Ӧֻ�����������ɫ����ʵ���з��ַ�̪��������ɫ���ɴ�������µIJ��룺
A��������Ư����
B������������Ư����
C��
[ʵ�����ж�] ��������б���
| ʵ���� | 1 | 2 | 3 |
| ʵ��װ�� |  |  |  |
| ��֤���� | | C | |
| ʵ������ | ��Һ������ɫ | ||
| ʵ��˵�� | 1��2��ʵ����NaOH��Һ���� ����������ƹ��塱���������ƹ��塱�����������ƹ��塱������ˮ���Ƶġ� | ||
��2����������ʵ��������֣�����������ˮ��Ӧ�����У���Ԫ���γ����ȶ��Ļ������Һ�л�������һ�ֲ����ȶ�������Ư���Ե�����X��X�Ļ�ѧʽ�� ��
��3��������ͼװ�ö���Һ�в����ȶ������ʽ���̽�����ڢٴ�װ��������� ����ѡ���ͬ�����ڴ�װ��������� ��

A����̪�Լ� B������������ˮ��Ӧ�����Һ
C���������� D���������ƹ������Ƶ���Һ
��4��������� ������������ˮ��Ӧ�Ļ�ѧ����ʽû��д��X����ԭ���� ��



 CuCl42-(��ɫ)+4H2O��
CuCl42-(��ɫ)+4H2O��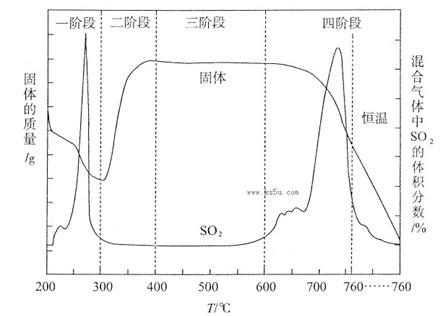



 BaCO3��+H2O)
BaCO3��+H2O)
