��Ŀ����
10���л���A��ͼ��ʾת����ϵ����A������ͼ���ʺɱ����ֵΪ88���������C��H��O����Ԫ�ص�������Ϊ6��1��4����A����ʹBr2��CCl4��Һ��ɫ��1mol B��Ӧ������2mol C��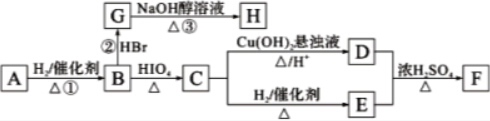
��֪��RCH��OH��-CH��OH��R��
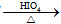 RCHO+R��CHO
RCHO+R��CHO��ش��������⣺
��1��A�Ľṹ��ʽΪ
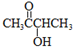 ��
����2�����١��ڡ���������Ӧ�IJ��ʷֱ�Ϊ90.0%��82.0%��75.0%������A�ϳ�H���ܲ���Ϊ55.4%��
��3��D+E��F�ķ�Ӧ����Ϊ������ȡ������Ӧ��
��4��д��C��������Һ��Ӧ�����ӷ���ʽΪCH3CHO+2[Ag��NH3��2]++2OH-
 CH3COO-+NH4++2Ag��+3NH3+H2O��
CH3COO-+NH4++2Ag��+3NH3+H2O����5����H����������̼ԭ�Ӳ���һ��ֱ���ϣ���H��һ�������ºϳ�˳���Ļ�ѧ����ʽΪ
 ����H����������̼ԭ�Ӿ���һ��ֱ���ϣ���Gת��ΪH�Ļ�ѧ����ʽΪ
����H����������̼ԭ�Ӿ���һ��ֱ���ϣ���Gת��ΪH�Ļ�ѧ����ʽΪ ��
����6���л���A�кܶ�ͬ���칹�壬��д��ͬʱ��������������һ���칹��X�Ľṹ��ʽ
 ��
��a��X�˴Ź���������3���壬�����֮��Ϊ1��1��2
b��1mol X����HIO4���ȵ������·�Ӧ�����γ�1mol ��Ԫȩ
c��1mol X�������2mol Na��Ӧ
d��X����NaHCO3��Ӧ��Ҳ����NaOH��Ӧ��Ҳ����Br2�����ӳɷ�Ӧ��
���� ����A������C��H��O����Ԫ�ص�������Ϊ6��1��4��֪C��H��O�����ʵ���֮��Ϊ2��4��1��������A������ͼ���ʺɱ����ֵΪ88������A�ķ���ʽΪC4H8O2�����ݷ���ʽ��֪A����һ�������Ͷȣ��ֲ���ʹBr2��CCl4��Һ��ɫ������A�д���C=O�����ٸ���1mol B��Ӧ������2mol C�������֪��������֪A�ĽṹΪ��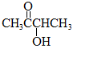 ���������Խ�һ���Ƴ�B��C��D��E��F�Ľṹ��ʽ�����������ǣ�
���������Խ�һ���Ƴ�B��C��D��E��F�Ľṹ��ʽ�����������ǣ�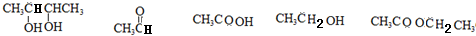 ������B��HBr����ȡ����Ӧ����±�������NaOH����Һ�з�����ȥ��Ӧ����H����������̼ԭ�Ӿ���һ��ֱ���ϣ����Կ����Ƶ�G��H�Ľṹ�ֱ�Ϊ��
������B��HBr����ȡ����Ӧ����±�������NaOH����Һ�з�����ȥ��Ӧ����H����������̼ԭ�Ӿ���һ��ֱ���ϣ����Կ����Ƶ�G��H�Ľṹ�ֱ�Ϊ��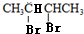 ��
��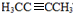 ���ݴ˿��Խ��⣮
���ݴ˿��Խ��⣮
��� �⣺����A������C��H��O����Ԫ�ص�������Ϊ6��1��4��֪C��H��O�����ʵ���֮��Ϊ2��4��1��������A������ͼ���ʺɱ����ֵΪ88������A�ķ���ʽΪC4H8O2�����ݷ���ʽ��֪A����һ�������Ͷȣ��ֲ���ʹBr2��CCl4��Һ��ɫ������A�д���C=O�����ٸ���1mol B��Ӧ������2mol C�������֪��������֪A�ĽṹΪ�� ���������Խ�һ���Ƴ�B��C��D��E��F�Ľṹ��ʽ�����������ǣ�
���������Խ�һ���Ƴ�B��C��D��E��F�Ľṹ��ʽ�����������ǣ�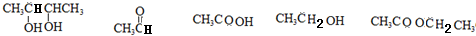 ������B��HBr����ȡ����Ӧ����±�������NaOH����Һ�з�����ȥ��Ӧ����H����������̼ԭ�Ӿ���һ��ֱ���ϣ����Կ����Ƶ�G��H�Ľṹ�ֱ�Ϊ��
������B��HBr����ȡ����Ӧ����±�������NaOH����Һ�з�����ȥ��Ӧ����H����������̼ԭ�Ӿ���һ��ֱ���ϣ����Կ����Ƶ�G��H�Ľṹ�ֱ�Ϊ��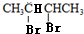 ��
��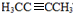 ��
��
��1������A������C��H��O����Ԫ�ص�������Ϊ6��1��4��֪C��H��O�����ʵ���֮��Ϊ2��4��1��������A������ͼ���ʺɱ����ֵΪ88������A�ķ���ʽΪC4H8O2�����ݷ���ʽ��֪A����һ�������Ͷȣ��ֲ���ʹBr2��CCl4��Һ��ɫ������A�д���C=O�����ٸ���1mol B��Ӧ������2mol C�������֪��������֪A�ĽṹΪ�� ��
��
�ʴ�Ϊ��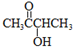
��2����A�ϳ�H���ܲ���ӦΪ�٣��ڡ���������Ӧ�IJ���֮����������A�ϳ�H���ܲ���=90.0%��82.0%��75.0%=55.4%��
�ʴ�Ϊ��55.4%��
��3���Ƚ�D��E��F�Ľṹ��֪��D+E��F�ķ�Ӧ����Ϊ������ȡ������Ӧ��
�ʴ�Ϊ��������ȡ������Ӧ��
��4��������ķ�����֪��CΪ��ȩ������������Һ��Ӧ�����ӷ���ʽΪ��CH3CHO+2[Ag��NH3��2]++2OH-  CH3COO-+NH4++2Ag��+3NH3+H2O��
CH3COO-+NH4++2Ag��+3NH3+H2O��
�ʴ�Ϊ��CH3CHO+2[Ag��NH3��2]++2OH-  CH3COO-+NH4++2Ag��+3NH3+H2O��
CH3COO-+NH4++2Ag��+3NH3+H2O��
��5����H����������̼ԭ�Ӳ���һ��ֱ���ϣ���HΪ1��3-����ϩ��H��һ�������ºϳ�˳���Ļ�ѧ����ʽΪ ������B��2��3-����������HBr����ȡ����Ӧ����±����
������B��2��3-����������HBr����ȡ����Ӧ����±����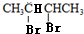 ������NaOH����Һ�з�����ȥ��Ӧ����H����H����������̼ԭ�Ӿ���һ��ֱ���ϣ����Կ����Ƶ�G��H�Ľṹ��GΪ
������NaOH����Һ�з�����ȥ��Ӧ����H����H����������̼ԭ�Ӿ���һ��ֱ���ϣ����Կ����Ƶ�G��H�Ľṹ��GΪ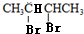 HΪ
HΪ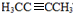 ��Gת��ΪH�Ļ�ѧ����ʽΪ
��Gת��ΪH�Ļ�ѧ����ʽΪ ��
��
�ʴ�Ϊ�� ��
�� ��
��
��6��A�ĽṹΪ�� ������������a��X�˴Ź���������3���壬�����֮��Ϊ1��1��2����X������λ�õ��⣬�Ҹ���֮��Ϊ1��1��2��b��1mol X����HIO4���ȵ������·�Ӧ�����γ�1mol ��Ԫȩ��������Ϣ��֪XӦΪ���������ǻ��Ļ�״�c��1mol X�������2mol Na��Ӧ��˵��X��2���ǻ���d��X����NaHCO3��Ӧ��Ҳ����NaOH��Ӧ��Ҳ����Br2�����ӳɷ�Ӧ��˵����Xû���Ȼ���������̼̼˫������X�ĽṹΪ
������������a��X�˴Ź���������3���壬�����֮��Ϊ1��1��2����X������λ�õ��⣬�Ҹ���֮��Ϊ1��1��2��b��1mol X����HIO4���ȵ������·�Ӧ�����γ�1mol ��Ԫȩ��������Ϣ��֪XӦΪ���������ǻ��Ļ�״�c��1mol X�������2mol Na��Ӧ��˵��X��2���ǻ���d��X����NaHCO3��Ӧ��Ҳ����NaOH��Ӧ��Ҳ����Br2�����ӳɷ�Ӧ��˵����Xû���Ȼ���������̼̼˫������X�ĽṹΪ ��
��
�ʴ�Ϊ�� ��
��
���� ���⿼�����л������ŵ����ʡ��л��ṹ���ƶϡ��л���Ӧ���͡�ͬ���칹�����д��֪ʶ���е��Ѷȣ�������ѵ��ڵڣ�6��С�ʣ�д��������ͬ���칹�壬��ѧ�������л��ṹ�ص㼰�����л�ȡ��Ϣ�������������Ҫ��ϸߣ�

 ��ͼͼ�麮����ҵ������ҵ���ִ�ѧ������ϵ�д�
��ͼͼ�麮����ҵ������ҵ���ִ�ѧ������ϵ�д�| A�� | Na+��CH3COO-��NH4+ | B�� | Mg2+��Ba2+��Br- | ||
| C�� | Cu2+��Cl-��I- | D�� | K+��Cl-��Mg2+ |
| A�� | �ձ�����Һת�Ƶ�����ƿ��ʱ��δϴ���ձ� | |
| B�� | ��Һʱ��������Һ�彦�� | |
| C�� | ����ʱ�����ӿ̶��� | |
| D�� | ����ҡ�Ⱥ���Һ���½���������ˮ���̶��� |
 2015��ŵ��������ѧ��ҽѧ�������ҹ�Ůҩѧ�������ϣ�������ҩ�з����������Ӧ����ű�����ƣ������صļ�-�߽ṹ��ʽ��ͼ�����й��������ص�˵������ȷ���ǣ�������
2015��ŵ��������ѧ��ҽѧ�������ҹ�Ůҩѧ�������ϣ�������ҩ�з����������Ӧ����ű�����ƣ������صļ�-�߽ṹ��ʽ��ͼ�����й��������ص�˵������ȷ���ǣ�������| A�� | ����ʽΪC15H22O5 | B�� | ���ڷ����廯���� | ||
| C�� | �ɷ���ˮ�ⷴӦ | D�� | ���й�������-O-O-����ǿ������ |

| A�� | ����ƿ����Һ©���͵ζ��ܶ���Ҫ��© | |
| B�� | ��װ�ü�Һ���ų�ˮ����ٴӷ�Һ©���¿ڷų��л��� | |
| C�� | ��װ���Ҽ��ȷֽ�NaHCO3���� | |
| D�� | ����500mL0.1mol•L?1NaCl��Һ�õ��IJ�������ֻ��500mL����ƿ���ձ��Ͳ����� |
| A�� | �����£�23g NO2��N2O4�Ļ�����庬��NA����ԭ�� | |
| B�� | 1L0.1mol•L-1�İ�ˮ����0.1NA��OH- | |
| C�� | ��״���£�NO��O2��11.2L��ϣ����û������ķ�������Ϊ0.75NA | |
| D�� | 1molFe��1molCl2��ַ�Ӧ��ת��3NA������ |
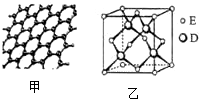 ԭ�������������������Ԫ��A��B��C��D��Eλ�����ڱ���ǰ�����ڣ�A��̬ԭ�ӵ�2p�������2��δ�ɶԵ��ӣ�C�������������Ǵ�����������3����C��Dͬ�������ڣ�Eλ�����ڱ���ds���������ֻ��һ�ԳɶԵ��ӣ���ش��������⣺
ԭ�������������������Ԫ��A��B��C��D��Eλ�����ڱ���ǰ�����ڣ�A��̬ԭ�ӵ�2p�������2��δ�ɶԵ��ӣ�C�������������Ǵ�����������3����C��Dͬ�������ڣ�Eλ�����ڱ���ds���������ֻ��һ�ԳɶԵ��ӣ���ش��������⣺