��Ŀ����
2�� ���������ḻ��ʣ�����Ľṹ�����ӣ��˽̰�γ̱����н̲ġ�ѡ��3��������չʾ��һЩ���;���Ľṹ�����ʵ����֪ʶ���磺�ɱ����������ʯ��ʯī���ǡ��ء�þ��ͭ��NaCl��CsCl��CaF2����ͼ��һ����������ͼ�������������������ʷ�Χ�ڻش��������⣺
���������ḻ��ʣ�����Ľṹ�����ӣ��˽̰�γ̱����н̲ġ�ѡ��3��������չʾ��һЩ���;���Ľṹ�����ʵ����֪ʶ���磺�ɱ����������ʯ��ʯī���ǡ��ء�þ��ͭ��NaCl��CsCl��CaF2����ͼ��һ����������ͼ�������������������ʷ�Χ�ڻش��������⣺��1����������ṹ�Ļ�����Ԫ�������������龧����Կ�����������ľ�����϶���ö��ɵģ�
��2��ij���Ӿ���ľ�����ͼ��B��C����������λ���÷��ӣ���ʾ����÷��Ӿ����Ǹɱ����÷��Ӿ����У�һ��������Χ����12�����ӣ�
��3����ͼ��NaCl�ľ���ͼ����A�����Na+����Cl-����B�����Cl-����Na+����C�����������
��4����ͼ�ǽ��ʯ�ľ���ͼ����̼ԭ�Ӵ���A���C��B���C��λ�ã�ѡ�A����B����C����
��5��ij��������ľ�����ͼ��A��C����������λ���ý���ԭ�ӣ���ʾ����ý���������ͭ����Cu����
���� ��1�����ݾ����Ķ��壬��������ṹ�Ļ�����Ԫ�������������龧����Կ�����������ľ�����϶���ö��ɵģ��ݴ˴��⣻
��2�����ݾ����Ľṹͼ��֪������λ��������Ķ�������ģ������������ṹ���ݴ��жϣ�
��3��NaCl�ľ�����ÿ����������Χ��6�������ӣ�ÿ����������Χ��6�������ӣ��ݴ��жϣ�
��4�����ʯ�ľ�����ÿ��̼ԭ����Χ��4��̼ԭ�ӣ��ݴ��жϣ�
��5�����ݽ�������ľ���ͼ��A��C����������λ���ý���ԭ�ӣ���֪��Bԭ��������λ�ã������������ѻ���ʽ���ݴ˴��⣻
��� �⣺��1�����ݾ����Ķ��壬��������ṹ�Ļ�����Ԫ�������������龧����Կ�����������ľ�����϶���ö��ɵģ�
�ʴ�Ϊ����������϶���ã�
��2�����ݾ����Ľṹͼ��֪������λ��������Ķ�������ģ������������ṹ�����������ʷ�Χ�ڿ�֪Ӧ�Ǹɱ����ɱ������У��Զ��������̼Ϊ�������������̼������Χ���ڵķ���λ�ھ����ö������������ϣ�����12����
�ʴ�Ϊ���ɱ���12��
��3��NaCl�ľ�����ÿ����������Χ��6�������ӣ�ÿ����������Χ��6�������ӣ�����A�����Na+��B�����Cl-����B�����Na+��A�����Cl-��C�����������
�ʴ�Ϊ��Na+���� Cl-����Cl- ����Na+���� ������
��4�����ʯ�ľ�����ÿ��̼ԭ����Χ��4��̼ԭ�ӣ�̼ԭ�Ӵ���A���C��B���C��
�ʴ�Ϊ��A���C��B���C��
��5�����ݽ�������ľ���ͼ��A��C����������λ���ý���ԭ�ӣ���֪��Bԭ��������λ�ã������������ѻ���ʽ�����Ըý���������ͭ����Cu����
�ʴ�Ϊ��ͭ����Cu����
���� ���⿼�鳣������ṹ�����ؿ���ѧ�������жϼ��ռ�������������Ϥ�������ʽṹ����������ע����ʯ��̼ԭ��������λ�ã���Ŀ�ѶȲ���

 ��ʦ�㲦��ϵ�д�
��ʦ�㲦��ϵ�д� Ӣ�żƻ���ĩ����ϵ�д�
Ӣ�żƻ���ĩ����ϵ�д���1��������0.1mol/L��CH3COOH��Һ��ˮϡ���̣����б���ʽ������һ����С����A��
A��c��H+�� B��$\frac{c��{H}^{+}��}{c��C{H}_{3}COOH��}$ C��c��H+��•c��OH-�� D��$\frac{c��O{H}^{-}��}{c��{H}^{+}��}$
��2����ͬ�����£�ȡ������ļס�������Һ����ϡ��100����ϡ�ͺ����Һ����pH��С��ϵΪ��pH���ף���pH���ң��� ���������������=������
��3����֪25��ʱ����ĵ���ƽ�ⳣ�����£�
| ��ѧʽ | CH3COOH | H2CO3 | HClO |
| ����ƽ�ⳣ��K1 | 1.8��10-5 | 4.3��10-7 | 3.0��10-8 |
| K2 | -- | 5.6��10-11 | -- |
CH3COOH+Na2CO3����������2CH3COOH+CO32-=H2O+2CH3COO-+CO2����
HClO+Na2CO3����������HClO+CO32-=ClO-+HCO3-��
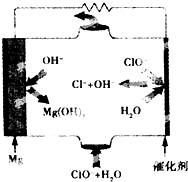 þȼ�ϵ�ؾ��б������ߡ�ʹ�ð�ȫ���㡢ԭ������Դ�ḻ���ɱ��͡�ȼ���������˼���ȾС���ص㣮��ͼΪþ--��������ȼ�ϵ�صĹ���ԭ��ͼ�������й�˵������ȷ���ǣ�������
þȼ�ϵ�ؾ��б������ߡ�ʹ�ð�ȫ���㡢ԭ������Դ�ḻ���ɱ��͡�ȼ���������˼���ȾС���ص㣮��ͼΪþ--��������ȼ�ϵ�صĹ���ԭ��ͼ�������й�˵������ȷ���ǣ�������| A�� | ���ܷ�ӦʽΪMg+ClO-+H2O=Mg��OH��2��+Cl- | |
| B�� | ������ӦʽΪClO-+H2O+2e-=Cl-+2OH- | |
| C�� | ��ȼ�ϵ����þΪ������������ԭ��Ӧ�� | |
| D�� | �ŵ������OH-���� |
| A�� | ��������Ϊ��̽��������ζ���ɴ��Լ�ƿ��ȡ��������Ʒ�� | |
| B�� | �Ȼ������弫������ˮ�������Ȼ���β��ʱӦ�������ӷ�����װ�� | |
| C�� | ����������ϡ������Һ����ȡ����Ũ�������Թ��м�ˮϡ�� | |
| D�� | �ƾ��Ʋ���ʱ�����������𣬼ȼ���ֿ�� |
| A�� | ����ʯ��ˮ������������Cl2+OH-�TCl-+ClO-+H+ | |
| B�� | ������Na������ˮ�У�Na+2H2O�T2Na++2OH-+H2�� | |
| C�� | ��ϡ�����м����������ۣ�3Fe+8H++2NO3�T3Fe2++2NO��+4H2O | |
| D�� | ̼�������Һ�м����������Լ���Һ��OH-+Ca2++HCO3-=H2O+CaCO3�� |
| A�� | ͨ�����ȵ�þ�۳�ȥN2�е�O2 | |
| B�� | ��ʪ��⻯�ص�����ֽ����Br2��g����NO2 | |
| C�� | ���ˮ�еμӱ���FeCl3��Һ�Ʊ�Fe��OH��3���� | |
| D�� | ������Һ���Ƿ���SO42-���Ƚ���Һ�������������ữ���ټ���BaCl2��Һ |
| A�� | ʹ�������Դ�Ƿ�ֹ���귢������Ҫ��ʩ֮һ | |
| B�� | CO2��CH4��N2�Ⱦ����������ЧӦ������ | |
| C�� | ���ܼ��ŷ��ϵ�̼���õ�Ҫ�� | |
| D�� | �����������ÿ�ȼ������̬����ˮ��������ڻ�����Դ��ȱ |
| A�� | �٣��ڣ��ۣ��� | B�� | �ۣ��٣��ܣ��� | C�� | �٣��ۣ��ܣ��� | D�� | �ڣ��ۣ��ܣ��� |
| A�� | Ũ���� | B�� | ��ʯ�� | C�� | ��ˮ�Ȼ��� | D�� | ���������� |