��Ŀ����
10�����¶�t���£�ijBa��OH��2��ϡ��Һ��c��H+��=10-amol/L��c��OH-��=10-bmol/L����֪a+b=12�������Һ����μ���pH=b��NaHSO4����û����Һ�IJ���pH���±���ʾ��| ��� | �������������/mL | �������Ƶ����/mL | ��Һ��pH |
| �� | 33.00 | 0.00 | 8 |
| �� | 33.00 | x | 7 |
| �� | 33.00 | 33.00 | 6 |
��2��b=4��x=27mL��
��3����Ӧ�۵����ӷ���ʽΪBa2++2OH-+2H++SO42-=BaSO4��+2H2O
��4�������¶��µ�Ba��OH��2��Һȡ��1mL����ˮϡ����1L����ϡ�ͺ���Һ��
c��Ba2+���sc��OH-��=1��20��
��5����NaHSO4��ͬ��NaHSO3 ��NaHCO3ҲΪ��ʽ�Σ���֪NaHSO3��Һ�����ԣ�NaHCO3��Һ�ʼ��ԣ�����Ũ�Ⱦ�Ϊ0.1mol/L��NaHSO3��Һ��NaHCO3��Һ����Һ�и����ӵ����ʵ���Ũ�ȴ������й�ϵ��R��ʾS��C�������п�����ȷ����ABC������ȷ�𰸵ı�ţ���
A��c��Na+����c��HRO${\;}_{3}^{-}$����c��H+����c��RO${\;}_{3}^{2-}$����c��OH-��
B��c��Na+��+c��H+��=c��HRO${\;}_{3}^{-}$��+2c��RO${\;}_{3}^{2-}$��+c��OH-��
C��c��H+��+c��H2RO3��=c��RO${\;}_{3}^{2-}$��+c��OH-��
D������Һ��c��Na+����c��HRO${\;}_{3}^{-}$����c��RO${\;}_{3}^{2-}$���ֱ���ȣ�
���� ��1������Kw=c��H+��•c��OH-������ˮ�����ӻ���Ȼ�����ˮ�ĵ������Ϊ���ȹ����ж��¶ȴ�С��
��2������ˮ�������ӻ�����Һ��pH���������������Һ������������Ũ�ȣ�����Һ��Ϻ���Һ��pH=8�����ݸ��¶���ˮ�����ӻ���֪�����Һ��ʾ���ԣ�˵�����������ӹ����������������ݼ����x��
��3����Ӧ�����Һ��pH=6����ҺΪ���ԣ�˵������ǡ�÷�Ӧ���ݴ�д����Ӧ�����ӷ���ʽ��
��4���ȸ�������������Һ������������Ũ�ȼ��������������Ũ�ȡ�������Ũ�ȣ��ټ����ϡ��1000������Һ�б�����Ũ�ȣ�����������������Һ�У�������������ϡ��������Ũ��ֻ�����ӽ�����ʱ����Ũ�ȣ��ݴ˼��������Ũ��֮�ȣ�
��5��A��NaHSO3��Һ������˵�������������ӵ������ˮ�⣻
B��������Һ�е���غ�����жϣ�
C��������Һ�е������غ�����жϣ�
D����Һ�е����ˮ��̶Ȳ�ͬ������Ũ�Ȳ�ͬ��
��� �⣺��1�����¶���ˮ�����ӻ�Ϊ��Kw=c��H+��•c��OH-��=10-a��10-b=10-��a+b��=1��10-12��1��10-14��ˮ�ĵ���Ϊ���ȹ��̣����Ը��¶�t�棾25�棬
�ʴ�Ϊ������1��10-12��
��2������������Һ��pH=8��ˮ�������ӻ�Ϊ1��10-12������Һ������������Ũ��Ϊ��c��OH-��=10-bmol/L=$\frac{1��1{0}^{-12}}{1��1{0}^{-8}}$mol/L=10-4mol/L������b=4��
����Һ��Ϻ���Һ��pH=7������ˮ�������ӻ�Ϊ1��10-12��֪��Һ��ʾ���ԣ���Ӧ�����Һ������������Ũ��Ϊ10-5mol/L����$\frac{1��1{0}^{-4}mol/L����33-x����1{0}^{-3}L}{��33+x����1{0}^{-3}L}$=10-5mol/L����ã�x=27��
�ʴ�Ϊ��4��27��
��3�����¶��£���Һ��pH=6��˵����Һ��ʾ���ԣ���Ӧ�����ӷ���ʽΪBa2++2OH-+2H++SO42-=BaSO4��+2H2O���ʴ�Ϊ��Ba2++2OH-+2H++SO42-=BaSO4��+2H2O��
��4������������Һ������������Ũ��Ϊ��10-4mol/L��������������Ũ��Ϊ5��10-5mol/L��c��Ba2+��=c[Ba��OH��2]=5��10-5mol/L��1mL������������Һϡ�͵�1Lʱ��������Ũ��Ϊ��$\frac{5��1{0}^{-5}}{1000}$mol/L=5��10-8mol/L��������������Ũ��ֻ�����ӽ�1��10-6mol/L������ϡ�ͺ���Һ��c��Ba2+���sc��OH-��=5��10-8mol/L��1��10-6mol/L=1��20��
�ʴ�Ϊ��1��20��
��5��A������������������Һ��NaHSO3��Һ������˵�������������ӵ������ˮ�⡢��Һ������Ũ�ȴ�СΪ��c��Na+����c��HSO3-������c��SO32-����c��OH-�������Ը�����Ũ�ȹ�ϵ���ܳ�������A��ȷ��
B����Һ�д��ڵ���غ�Ϊ��c��Na+��+c��H+��=c��HRO3-��+2c��RO32-��+c��OH-������B��ȷ��
C�����ݵ���غ㣬c��Na+��+c��H+��=c��HRO3-��+2c��RO32-��+c��OH-���������غ�õ�c��Na+��=c��HRO3-��+c��RO32-��+c��H2RO3�������߽�ϵõ���c��H+��+c��H2RO3��=c��RO32-��+c��OH-������C��ȷ��
D����Һ�е����ˮ��̶Ȳ�ͬ����Һ�и�����Ũ�Ȳ�ͬ����D����
�ʴ�Ϊ��ABC��
���� ���⿼������Һ������Ũ�ȴ�С�Ƚϡ�ˮ�ĵ��뼰��Ӱ�����ء���Һ���������ҺpH�ļ����֪ʶ����Ŀ�Ѷ��еȣ������漰�������ϴ�֪ʶ��ϴ�ע�����������ʱ�Ķ����жϡ���Һ���������ҺpH�ļ��㷽������ȷ��Һ������Ũ�ȴ�С�Ƚϵķ�������4��Ϊ�״��㣬ע����ȷ����������Һϡ�ͺ�����������Ũ��Ϊ���¶��µ�����ʱ��Ũ�ȣ�

 �ŵ������ϵ�д�
�ŵ������ϵ�д� ijʵ��С�������50mL 1.0mol/L�����50mL 1.1mol/L����������Һ����ͼװ���н����кͷ�Ӧ���ڴ��ձ��ײ�����ĭ���ϣ���ֽ������ʹ�����С�ձ���������ձ�������ƽ��Ȼ�����ڴ�С�ձ�֮����������ĭ���ϣ���ֽ���������ձ�������ĭ���ϰ壨��Ӳֽ�壩���ǰ壬�ڰ��м俪����С�ף�����ʹ�¶ȼƺͻ��β��������ͨ����ͨ���ⶨ��Ӧ���������ų��������ɼ����к��ȣ��Իش��������⣺
ijʵ��С�������50mL 1.0mol/L�����50mL 1.1mol/L����������Һ����ͼװ���н����кͷ�Ӧ���ڴ��ձ��ײ�����ĭ���ϣ���ֽ������ʹ�����С�ձ���������ձ�������ƽ��Ȼ�����ڴ�С�ձ�֮����������ĭ���ϣ���ֽ���������ձ�������ĭ���ϰ壨��Ӳֽ�壩���ǰ壬�ڰ��м俪����С�ף�����ʹ�¶ȼƺͻ��β��������ͨ����ͨ���ⶨ��Ӧ���������ų��������ɼ����к��ȣ��Իش��������⣺��1�����β����������������ʹ��Һ���»�Ͼ��ȣ���֤��Һ���µ��¶ȴﵽһ�£�
��2����ʵ�������Թ�����NaOH��ԭ��̲���˵��Ϊ��֤������ȫ���кͣ����ʣ������ڷ�Ӧ������Ϊ�з�������������������ڷ�Ӧ�лӷ������õ��к���ƫС���ƫ��ƫС�����䡱����
��3����ʵ��С����������ʵ�飬ÿ��ȡ��Һ��50mL������¼����ԭʼ���ݣ�?
| ʵ����� | ��ʼ�¶�t1/�� | ��ֹ�¶�t2/�� | �²t2-t1��/�� | ||
| ���� | NaOH��Һ | ƽ��ֵ | |||
| 1 | 25.1 | 24.9 | 25.0 | 31.6 | 6.6 |
| 2 | 25.1 | 25.1 | 25.1 | 30.6 | 5.5 |
| 3 | 25.1 | 25.1 | 25.1 | 31.9 | 6.8 |
��4�����õ�Ũ�ȵĴ�����NaOH��Һ��Ӧ�����õ��к��Ȼ�ƫ���ƫ��ƫС�����䡱������ԭ�����ô���������ᣬ�������Ҫ������������ɲ�õ��к�����ֵƫС�����к���Ϊ��ֵ�������к���ƫ��?

| A�� | ʵ����ư��� | |
| B�� | ʵ���������Ũ�����ᾧ | |
| C�� | ʵ�������1��1��ϡ���� | |
| D�� | ʵ�������KMnO4����Һ�ζ�δ֪Ũ�ȵ�Na2SO3��Һ |

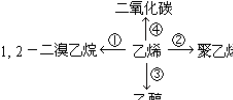
 ��CH2=CH2+H2O$\stackrel{һ������}{��}$CH3CH2OH��CH2=CH2+3O2$\stackrel{��ȼ}{��}$2CO2+2H2O��
��CH2=CH2+H2O$\stackrel{һ������}{��}$CH3CH2OH��CH2=CH2+3O2$\stackrel{��ȼ}{��}$2CO2+2H2O��
 2C��g��+B��g����H=+150akJ/mol��
2C��g��+B��g����H=+150akJ/mol��

 ��
�� ��ΪC��ͬϵ�д����������������X��һ��ͬ���칹��Ľṹ��ʽ��
��ΪC��ͬϵ�д����������������X��һ��ͬ���칹��Ľṹ��ʽ�� ��
�� ��
�� ������һ�ּ��ɣ���
������һ�ּ��ɣ��� ��
�� Ϊԭ���Ʊ�
Ϊԭ���Ʊ� �ĺϳ�·������ͼ�����Լ����ã����ϳ�·������ͼʾ�����£�
�ĺϳ�·������ͼ�����Լ����ã����ϳ�·������ͼʾ�����£�