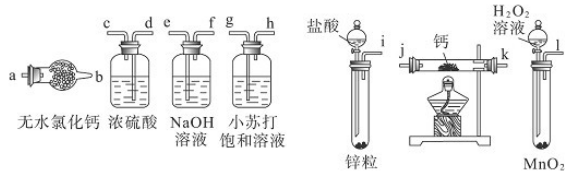
��Ŀ����
ijͬѧ���������ʵ�飬�ԲⶨCaCO3��(NH4)2CO3��NH4Cl���������е�Ԫ�ص�����������ʵ���������ͼ��ʾ��
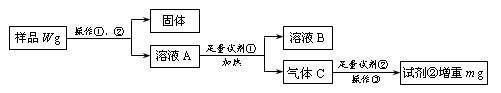
��ش�:
��1��Ϊ���в����١��ڣ���Ҫ�õ��IJ���������_____________________��
��2��ԭ��Ʒ�е�Ԫ�ص���������Ϊ_________________���ú�W��m�Ĵ���ʽ��ʾ����
��3������ͬѧ���ڲ���������ʹ�ⶨ���ƫ�ͣ���ԭ�������_________������ĸ����
a. �Լ����������� b. ����δ��ϴ��
c. �Լ����������� d. ����Cδ������

��ش�:
��1��Ϊ���в����١��ڣ���Ҫ�õ��IJ���������_____________________��
��2��ԭ��Ʒ�е�Ԫ�ص���������Ϊ_________________���ú�W��m�Ĵ���ʽ��ʾ����
��3������ͬѧ���ڲ���������ʹ�ⶨ���ƫ�ͣ���ԭ�������_________������ĸ����
a. �Լ����������� b. ����δ��ϴ��
c. �Լ����������� d. ����Cδ������
��1���ձ���©��������������3�֣���2��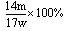 ��1�֣�
��1�֣�
��3��a��b��c ����3�֣�
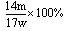 ��1�֣�
��1�֣� ��3��a��b��c ����3�֣�
��

��ϰ��ϵ�д�
 ����������ϵ�д�
����������ϵ�д� �Ż���ҵ�Ϻ��Ƽ����׳�����ϵ�д�
�Ż���ҵ�Ϻ��Ƽ����׳�����ϵ�д�
�����Ŀ
 _������Һת��________�У�ϴ�ӣ����ݣ�ҡ�ȡ�
_������Һת��________�У�ϴ�ӣ����ݣ�ҡ�ȡ� ��ܡ����ܡ�������1��0 mol��L��1 Ba��OH��2��Һ��
��ܡ����ܡ�������1��0 mol��L��1 Ba��OH��2��Һ��
 ����
����
 ��2���Ӵ���������Һ��pH=5~6���������Լ5~10���ӣ���������Ҫ��MnO2�������ֳ����������ֳ����� ��д��ѧʽ�������д���������Һ��MnSO4ת��ΪMnO2�����ӷ���ʽΪ�� ��
��2���Ӵ���������Һ��pH=5~6���������Լ5~10���ӣ���������Ҫ��MnO2�������ֳ����������ֳ����� ��д��ѧʽ�������д���������Һ��MnSO4ת��ΪMnO2�����ӷ���ʽΪ�� �� ��= ��
��= ��