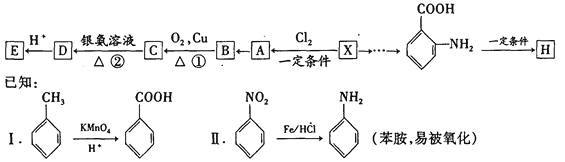
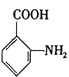
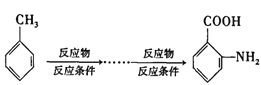
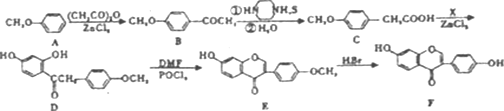
��Ŀ����
����Ŀ����ͼ��ʾװ���У��ס��ҡ��������ձ����ηֱ�ʢ��100 g 5.00%��NaOH��Һ��������CuSO4��Һ��100 g 10.00%��K2SO4��Һ���缫��Ϊʯī�缫��
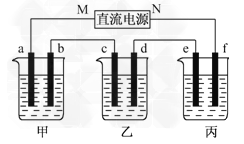
(1)��ͨ��Դ������һ��ʱ���ñ���K2SO4Ũ��Ϊ10.47%������c�缫�������ӡ��ݴ˻ش����⣺
����Դ��N��Ϊ____________________����
���缫b�Ϸ����ĵ缫��ӦΪ____________________��
����ʽ����缫b�����ɵ������ڱ�״���µ������________________��
���缫c�������仯��__________g��
�����ǰ�����Һ���ᡢ���Դ�С�Ƿ����仯��������ԭ����Һ______________________________������Һ______________________________������Һ______________________________��
(2)�����������ͭȫ����������ʱ����ܷ�������У�Ϊʲô�� ________________________________________��
���𰸡� �� 4OH����4e��===2H2O��O2�� ˮ���ٵ�������100g��(1��![]() )��4.5g ����O2�����
)��4.5g ����O2����� ![]() ��
��![]() ��22.4L��mol-1=2.8L 16 ����������Ϊ����ˮ�����٣���Һ��NaOHŨ������ ����������Ϊ������OH������O2����Һ��H������Ũ������ ����Դ�Сû�б仯����ΪK2SO4��ǿ��ǿ���Σ�Ũ�����Ӳ�Ӱ����Һ������� �ܼ������У���ΪCuSO4��Һ��ת��ΪH2SO4��Һ����ӦҲ�ͱ�Ϊˮ�ĵ�ⷴӦ
��22.4L��mol-1=2.8L 16 ����������Ϊ����ˮ�����٣���Һ��NaOHŨ������ ����������Ϊ������OH������O2����Һ��H������Ũ������ ����Դ�Сû�б仯����ΪK2SO4��ǿ��ǿ���Σ�Ũ�����Ӳ�Ӱ����Һ������� �ܼ������У���ΪCuSO4��Һ��ת��ΪH2SO4��Һ����ӦҲ�ͱ�Ϊˮ�ĵ�ⷴӦ
��������(1)���ұ���c�������ӣ�˵��Cu������c�缫�ϣ������Ǵ�b-c�ƶ���M�Ǹ�����NΪ������
������ΪNaOH���൱�ڵ��H2O������b��Ϊ������OH-�ŵ磬��4OH--4e-=2H2O+O2����
������ΪK2SO4���൱�ڵ��ˮ�������ˮ������Ϊx���ɵ��ǰ��������������У�100��10%=(100-x)��10.47%����x=4.5g����Ϊ0.25mol���ɷ���ʽ2H2+O2�T2H2O��֪������2molH2O��ת��4mol���ӣ�����������Ӧ��ת��0.5mol���ӣ�������O2Ϊ0.5/4=0.125mol������µ����Ϊ0.125��22.4=2.8L��
��������·�Ǵ����ģ�����ÿ���ձ��еĵ缫��ת�Ƶ���������ȵģ����ݵ缫��Ӧ��Cu2++2e-=Cu����֪ת��0.5mol�������ɵ�m(Cu)= ![]() ��64=16g��
��64=16g��
�������൱�ڵ��ˮ����NaOH��Ũ������pH�����������ΪCu2+�ŵ磬����ΪOH-�ŵ磬��ⷽ��ʽΪ��2CuSO4+2H2O![]() 2Cu+O2��+2H2SO4������H+���࣬��pH��С������Ϊ���ˮ������K2SO4���ԣ���pH�������䣻
2Cu+O2��+2H2SO4������H+���࣬��pH��С������Ϊ���ˮ������K2SO4���ԣ���pH�������䣻
(2)��ͭȫ������ʱ����Һ�����е�������ᣬ���Լ�����⣬�ʴ�Ϊ�����ԣ� ͭȫ�����������Լ������H2SO4���е��Һ���ɵ�⡣

 ����ѧҵ���Ե�����ϵ�д�
����ѧҵ���Ե�����ϵ�д�