��Ŀ����
ʵ���Һϳ����������ֲ�Ʒ�IJ������£�
��������ƿ�ڽ��������Ҵ�������ŨH2SO4��ϣ�Ȼ��Һ©���ߵμӴ��ᣬ��������ֱ���ռ������Ʒ�ɵõ������Ҵ������ѡ����ᡢ����ˮ�����������ֲ�Ʒ���ݴ˻ش����⣺
��1����Ӧ�м�����Ҵ��ǹ����ģ���Ŀ����
________________________________��
��2���ߵμӴ��ᣬ���������Ŀ����
_________________________________��
��3����ʵ����ʹ�����ιܳ������������⣬��һ��Ҫ������___________��
��4���Թ��б���̼������Һ��������___________________�� _______________��_____________________________��
��5��ʵ�����ɵ��������������ܶȱ�ˮ_______�������С��������_________ ��ζ��
��6��ʵ�������¶ȹ��ߣ���170�棩������������Ҫ�л�������__________�������ƣ���
��1�� ����һ�ַ�Ӧ��������ʹƽ��������Ӧ�����ƶ���1�֣�
��2������������ʹƽ��������Ӧ�����ƶ���1�֣�
��3�������� ��1�֣�
��4���ܽ��Ҵ���1�֣� �� �к����ᣨ1�֣� �� ���������������ܽ�ȣ�1�֣���
��5��С��1�֣��� ���㣨1�֣�
��6����ϩ��2�֣�
����:��

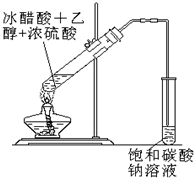 ʵ���Һϳ����������ֲ�Ʒ�ļ���װ�����ң��ɸ�ʵ����Եõ����������ֲ�Ʒ���ݴ���գ�
ʵ���Һϳ����������ֲ�Ʒ�ļ���װ�����ң��ɸ�ʵ����Եõ����������ֲ�Ʒ���ݴ���գ�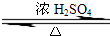 CH3COOCH2CH3+H2O��������Ӧ
CH3COOCH2CH3+H2O��������Ӧ ʵ���Һϳ����������ֲ�Ʒ�IJ������£���������ƿ�ڽ��������Ҵ�������Ũ�����ϣ�Ȼ��Һ©���ߵμӴ����������ֱ���ռ������Ʒ�ɵõ������Ҵ������ѡ����ᡢ����ˮ�����������ֲ�Ʒ���ݴ˻ش����⣺
ʵ���Һϳ����������ֲ�Ʒ�IJ������£���������ƿ�ڽ��������Ҵ�������Ũ�����ϣ�Ȼ��Һ©���ߵμӴ����������ֱ���ռ������Ʒ�ɵõ������Ҵ������ѡ����ᡢ����ˮ�����������ֲ�Ʒ���ݴ˻ش����⣺