��Ŀ����
[��ѧ��ѡ��3�����ʽṹ������]Ŀǰ�뵼���������ڽ���һ����ͭоƬ���������ڹ�оƬ����ͭ���������ߡ����ϵĽ���ͭ���ִ��Ƽ�Ӧ����ȡ����ͻ�ơ��û�ͭ��(��Ҫ�ɷ�ΪCuFeS2)������ͭ���䷴Ӧԭ�����£�
��1����̬ͭԭ�ӵļ۵����Ų�ʽΪ____________������Ԫ����ȣ���һ�����ܽϴ��Ԫ����________(��Ԫ�ط���)��
��2����Ӧ�١����о���������ͬ��������ӣ��÷��ӵ�����ԭ���ӻ�������_________��������ṹ��________����÷��ӻ�Ϊ�ȵ�����ĵ�������Ļ�ѧʽ�� ��
��3��ijѧ��������ͭ��Һ�백ˮ����һ��ʵ�飺CuSO4��Һ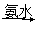 ��ɫ����
��ɫ����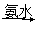 �����ܽ⣬�õ�����ɫ����Һ��������ɫ�������ڰ�ˮ�����ӷ���ʽΪ ��
�����ܽ⣬�õ�����ɫ����Һ��������ɫ�������ڰ�ˮ�����ӷ���ʽΪ ��
��4��ͭ�ǵ�����������Ҫ�Ĺ���Ԫ��֮һ���䵥�ʼ���������й㷺��;��ͭ������ͭԭ�Ӷѻ�ģ��Ϊ________��ͭ��ij�������ᄃ���ṹ��ͼ��ʾ�����þ�����ܶ�Ϊd g/cm3�������ӵ�������ֵΪNA����þ�����ͭԭ������ԭ��֮��ľ���Ϊ________pm��(�ú�d��NA��ʽ�ӱ�ʾ)

 �ŵ������ϵ�д�
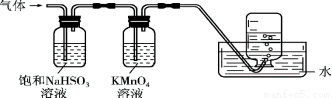
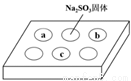
�ŵ������ϵ�д�ijͬѧ����SO2������ʵ�顣�ڵ�ΰ�a��b��c���ֱ���в�ͬ���Լ�������Na2SO3�����ϵμ�����ŨH2SO4����������ΰ��ϸ���������һ��ʱ���۲쵽��ʵ���������±���ʾ��
��� | �Լ� | ʵ������ |
|
a | Ʒ����Һ | ��ɫ��ȥ | |
b | ����KMnO4��Һ | ��ɫ��ȥ | |
c | NaOH��Һ����2�η�̪�� | ��ɫ��ȥ |
����˵����ȷ����
A. Ũ������Na2SO3���巢����������ԭ��Ӧ
B. a��b������SO2����Ư����
C. c��ֻ���ܷ�����Ӧ��SO2+ 2OH��= SO32��+ H2O
D. c��������Һ������Ũ��һ�����ڹ�ϵ��c(Na+)+c(H+) = 2c(SO32��) +c(HSO3?) + c(OH?)


 N2O4(g) ��H=-c kJ/mol
N2O4(g) ��H=-c kJ/mol
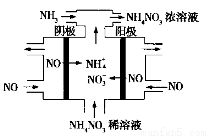
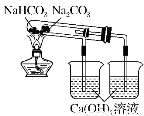
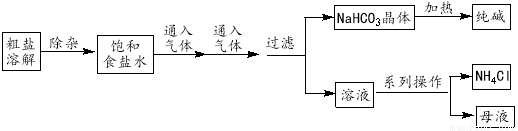
 HCO3��������ȶ���
HCO3��������ȶ��� CH3OH(g) ∆H1=?90.1kJ��mol?1
CH3OH(g) ∆H1=?90.1kJ��mol?1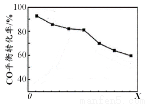
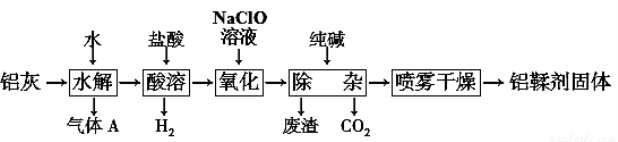
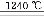 2Al2O3+2NH3��+N2��+5SO3��+3SO2��+53H2O
2Al2O3+2NH3��+N2��+5SO3��+3SO2��+53H2O