��Ŀ����
20����ȩ��һ����Ҫ�Ļ�����Ʒ�������ü״��������Ʊ�����̬��ȩ��HCHO������̬�״���CH3OH��ת����������ϵ��ͼ��ʾ����1���״�������ת��Ϊ��ȩ�ķ�Ӧ�����ȣ�����ȡ����ȡ�����Ӧ�����̢��У��÷�Ӧ�Ļ�ܵ�=��E4-E1��KJ/mol���ú�E�Ĵ���ʽ��ʾ����
��2�����̢�����̢�Ƚϣ��ı�������Ǽ����˴��������̢�����̢�ķ�Ӧ���Ƿ���ͬ����ͬ��ԭ����һ����ѧ��Ӧ�ķ�Ӧ�Ƚ��뷴Ӧ����ʼ״̬�ͷ�Ӧ������״̬�йأ����뷴Ӧ��;����
��3��д���״�������ת��Ϊ��ȩ���Ȼ�ѧ����ʽ��CH3OH��g��=HCHO��g��+H2��g����H=��E2-E1�� kJ/mol
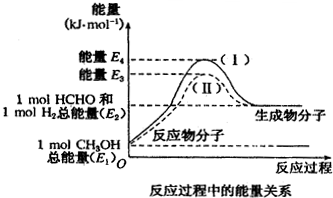
���� ��1����ͼ��֪����Ӧ��������С����������������
��2���ı��������ͷ�Ӧ�Ļ�ܣ���ʼ״̬���䣬�ʱ䲻�䣻
��3���״�������ת��Ϊ��ȩ����֪�״���Ӧ���ɼ�ȩ�����������ͼ�������仯�����ʱ䣬�Դ���д�Ȼ�ѧ����ʽ��
��� �⣺��1��ͨ��ͼ����Կ�������Ӧ�������������������������ͣ���Ϊ���ȷ�Ӧ���ʴ�Ϊ�����ȣ�
��2��һ����ѧ��Ӧ����ЧӦ���뷴Ӧ����ʼ״̬�ͷ�Ӧ������״̬�йأ��뷴Ӧ��;���أ���ȡ���ڷ�Ӧ�����������������Ĵ�С�����̢�����̢�ķ�Ӧ������ͬ�ģ��ı������Ϊ������
�ʴ�Ϊ�������˴�������ͬ��һ����ѧ��Ӧ�ķ�Ӧ�Ƚ��뷴Ӧ����ʼ״̬�ͷ�Ӧ������״̬�йأ����뷴Ӧ��;���أ�
��3����ͼ��֪���״�������ת��Ϊ��ȩΪ���ȷ�Ӧ��������������Ϊ��E2-E1�������Ȼ�ѧ����ʽΪCH3OH��g��=HCHO��g��+H2��g����H=+��E2-E1�� kJ/mol��
�ʴ�Ϊ��CH3OH��g��=HCHO��g��+H2��g����H=+��E2-E1�� kJ/mol��
���� ���⿼���Ȼ�ѧ����ʽ����д��Ϊ��Ƶ���㣬����ͼ�������仯���ʱ䡢�Ȼ�ѧ����ʽ����д����Ϊ���Ĺؼ������ط�����Ӧ�������Ŀ��飬��Ŀ�ѶȲ���

| ������ | K+��NH4+��Fe3+��Ba2+ |
| ������ | Cl-��Br-��CO32-��HCO3-��SO32-��SO42- |

��֪����ɫ����DΪ��һ���壮 ��ش��������⣺
��1������BΪNH3���ѧʽ������ɫ����EΪCaCO3���ѧʽ����
��2������Һ�п϶����ڵ���������HCO3-��CO32-��SO42-��
��3��д�����ɰ�ɫ����B�����ӷ���ʽ��HCO3-+Ba2++OH-=BaCO3��+H2O��
��4���ж�ԭ��Һ��K+�Ƿ���ڣ������ڣ������ʵ���Ũ�ȵ���СֵΪ���٣��������ڣ���˵�����ɣ����ڣ��ɵ���غ��֪K+������СŨ��Ϊ0.1mol/L��
| A�� | ��ȩ�ĵ���ʽ�� | B�� | �Ҵ����ӵ����ģ�ͣ� | ||
| C�� |  �����ƣ�3-��-2-��ϩ �����ƣ�3-��-2-��ϩ | D�� | CO2�ķ���ģ��ʾ��ͼΪ�� |
| �� | H+ | Na+ | A- |
| Ũ�ȣ�mol/L�� | 2.50��10-10 | 0.100 | 9.92��10-2 |
| A�� | ������Һ�У�c��Na+����c��A-����c��HA����c��OH-�� | |
| B�� | t��ʱ��ˮ�����ӻ�Kw��1.0��10-14 | |
| C�� | t��ʱ��һԪ��HA�ĵ��볣��K=3.10��10-8 | |
| D�� | t��ʱ��0.100mol/LNaA��Һ��A-��ˮ����Ϊ0.400% |
| A�� | �����Ƿ������绹�ǻ������磬���ǽ���ѧ��ת��Ϊ���� | |
| B�� | PM2.5���е�Ǧ���ӡ�����������ȶ������к���Ԫ�ؾ��ǽ���Ԫ�� | |
| C�� | ���������͵������������γ��������Ҫ���� | |
| D�� | ������ϴ�Ӽ������ƹ�ʹ�ã�������Ч����ˮ�帻Ӫ�����ķ��� |
| A�� | �ְ��е������������缫��ӦΪFe-2e-=Fe2+ | |
| B�� | �ŵ�ʱ���������������� | |
| C�� | �ŵ�ʱOH-�������ƶ� | |
| D�� | ����ܷ�ӦΪ��2Li+2H2O�T2LiOH+H2�� |
| A�� | FeCl3��Һ��ʴӡˢ��·ͭ�壺Cu+Fe3+�TCu2++Fe2+ | |
| B�� | �����ܽ⼦���ǣ�2H++CaCO3�TCa2++CO2��+H2O | |
| C�� | ��NaHSO4��Һ�е���Ba��OH��2��Һ����Һ�����ԣ�Ba2++2OH-+2H++SO42-�TBaSO4��+2H2O | |
| D�� | ��NaHCO3��Һ�е�����������ʯ��ˮ��HCO3-+Ca2++OH-�TCaCO3��+H2O |
| ����� | �Լ� | ���뷽�� | |
| A | �������ӣ� | ��ˮ | ���� |
| B | ���飨��ϩ�� | ���� | ϴ�� |
| C | �������������ᣩ | ����Na2CO3��Һ | ��Һ |
| D | �屽���嵥�ʣ� | ����ˮ | ���� |
| A�� | A | B�� | B | C�� | C | D�� | D |
 ��
�� ��A��ˮ�ڴ��������·����ӳɷ�Ӧ�Ļ�ѧ����ʽ
��A��ˮ�ڴ��������·����ӳɷ�Ӧ�Ļ�ѧ����ʽ ��ֻд��Ҫ�����
��ֻд��Ҫ�����