��Ŀ����
�����й���Һ��������ȷ����
| A�������µĴ������Һ�����ԣ�����Һ��c(H+)��c(OH-)��10-7mol��L-1 |
| B����pH��2�Ĵ�����Һ�м�������c(��)��2mol��L-1��ij����Һ�����Һ��pHһ�����С |
| C��pH��ͬ�Ĵ�����Һ�����ᣬ�ֱ�������ˮϡ����ԭ��Һ��m����n������ϡ�ͺ�����Һ��pH����ͬ����m��n |
| D��������ǿ����Һ�������ƶ�������Ŀһ���ȵ�����������Һ�������ƶ�������Ŀ�� |
A C
�����µĴ������Һ��笠����Ӻʹ�������Ӿ���ˮ�⣬���Ƿֱ�ˮ��õ���H+��OH-��Ũ����ȣ�����Һ�����ԣ���ʱ������ˮ�����ӻ�û��c(H+)��c(OH-)=10-7mol��L-1��B�����������������Աȴ��ỹҪ�������п���ʹ��Һ��pH������Һ�ĵ�����ȡ������Һ�������ƶ����ӵ�Ũ�ȶ�����Ŀ��D�����

��ϰ��ϵ�д�
�����Ŀ
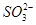 ��
�� ��
��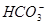 ��OH-��
��OH-�� ,��������������º����ܴ�������Һ�С�
,��������������º����ܴ�������Һ�С�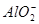 ʱ,���ܴ���___________________________________________________��
ʱ,���ܴ���___________________________________________________��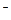 )]2=KSP������KSPΪ��������Ϊ���¶���Mg(OH)2���ܶȻ����������ƶ�������þ�����������е��ܽ���ɴ�С��˳����
)]2=KSP������KSPΪ��������Ϊ���¶���Mg(OH)2���ܶȻ����������ƶ�������þ�����������е��ܽ���ɴ�С��˳���� 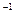 AlCl3��Һ ��0.1mol��L
AlCl3��Һ ��0.1mol��L