��Ŀ����
��֪A��B��C��D����ѧ��ѧ�г��������ֲ�ͬ���ӣ�����֮�������ͼ��ʾ��ת����ϵ����Ӧ�����Ѿ���ȥ����
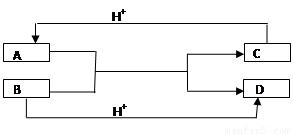
��1�� ���A��B��C��D����10���ӵ����ӣ���д��A��D�Ļ�ѧʽ��
A____________ D_______________
��2�����A��C��18���ӵ����ӣ�B��D��10���ӵ����ӣ���д����
�� A��B����Һ�з�Ӧ�����ӷ���ʽΪ��___________________________________
�� �����������ӷ���ʽ�������ж�C��B������ӵ�������С�ǣ��û�ѧʽ�����ӷ��ű�ʾ��_________________________________________________��
��1��A ��NH4�� D ��H2O
��2���� HS����OH����S2����H2O (��H2S��2OH����S2����2H2O��
�� OH����HS�� ����OH�� ��S2����
��������
�����������1�����A��B��C��D����10���ӵ����ӣ����10���������з����жϣ�10��������
һ�ˣ�Ne��N3-��O2-��F-��Na+��Mg2+��Al3+ �����ˣ�HF��OH-�����ˣ�H2O��NH2-���ĺˣ�NH3��H3O+ ��
��ˣ�CH4��NH4+�����ݷ�����ӦA+B��C+D��A��B��C��D����10���ӵ����ӣ����ж�Ϊ��A��B�ֱ�ΪOH����NH4+��C��D�ֱ�ΪNH3��H2O������ΪB�������ӷ�Ӧ����D������B��OH����D��ˮ��A��NH4+��C�ǰ����������������ӷ�Ӧ����NH4+��
��2�����A��C��18���ӵ����ӣ�B��D��10���ӵ����ӣ�����ݷ�ӦʽA+B��C+D��֪������������HS����OH����S2����H2O��H2S��2OH����S2����2H2O������ΪB�������ӷ�Ӧ����D������B��OH����D��ˮ��A��HS����H2S��C��S2����
�� A��B����Һ�з�Ӧ�����ӷ���ʽΪHS����OH����S2����H2O (��H2S��2OH����S2����2H2O��
�ڸ��ݷ���ʽHS����OH����S2����H2O (��H2S��2OH����S2����2H2O����֪��C��B������ӵ�������С��OH����HS�� ����OH����S2������
���㣺��������ͼ���10�����Լ�18���������ж�
�����������ۺ���ǿ����ѧ����˼ά����Ҫ��ߣ�����������ѧ������˼ά�����ͷ�ɢ˼ά������������Ҫ�ǿ���ԭ�Ӻ�����ӽṹ����������ת����ϵ��Ӧ�ã���Ҫ��10���Ӻ�18�������ķ�����Ӧ�á��������ճ���10���Ӻ�18�������ǽ���ؼ���

 ���Ͱ�ͨ��ĩ���ϵ�д�
���Ͱ�ͨ��ĩ���ϵ�д�| A��A��ԭ�Ӱ뾶��B��С | B��B��C�γɵĻ�����ֻ��һ�� | C��C�ڻ������г�+1�� | D��D���ʵľ���������뵼����� |
 ��B��ͬ������ԭ�ӵ�һ��������С��Ԫ�أ�Cԭ�ӵ������������δ�ɶԵ��ӣ�E�������ճ���������õĽ�����
��B��ͬ������ԭ�ӵ�һ��������С��Ԫ�أ�Cԭ�ӵ������������δ�ɶԵ��ӣ�E�������ճ���������õĽ�����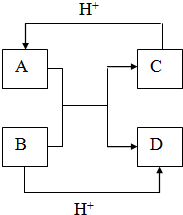 ��֪A��B��C��D����ѧ��ѧ�г��������ֲ�ͬ��������֮�������ͼ��ʾ��ת����ϵ��
��֪A��B��C��D����ѧ��ѧ�г��������ֲ�ͬ��������֮�������ͼ��ʾ��ת����ϵ��