��Ŀ����
����������ȷ����
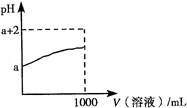
A��������pH=7��CH3COOH��CH3COONa�Ļ��Һ�����ӵ�Ũ�ȴ�С˳��Ϊ��
c��Na+��> c��CHCOO����> c��H+��= c��OH����
B��0.1 mol/L KHS����Һ�У�c��K+��= 2c��S2����+ c��HS����+ c��H2S��
C��25�� ʱ��pH��Ϊ12��NaOH��Һ��Na2CO3��Һ����ˮ�������c��OH������ǰ��С�ں���
D���к�pH�������ͬ�������������Һ������NaOH�����ʵ������
C

��ϰ��ϵ�д�
�����Ŀ
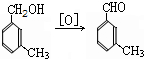
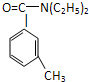 ���ð�������DEET����һ�ֶ��˰�ȫ�����Ը�����ҩ�Ե��������ü�����ṹ��ʽΪ����֪��RCOOH
���ð�������DEET����һ�ֶ��˰�ȫ�����Ը�����ҩ�Ե��������ü�����ṹ��ʽΪ����֪��RCOOH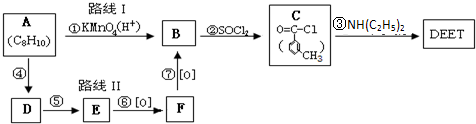
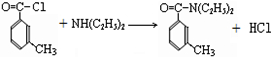
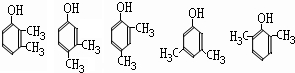 ����д2�֣�
����д2�֣�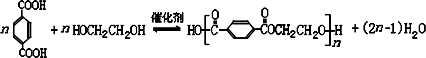
 NH3?H2O+H+
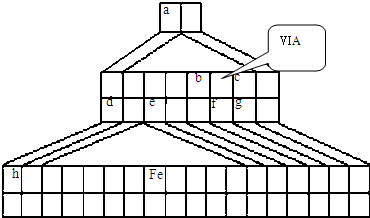
NH3?H2O+H+ ��2010?��������ģ��X��Y��Z��W��Ϊ����10���ӵ���������X��Y��ZΪ���ӣ�WΪ���ӣ���X��Z�����к��еĹ��õ��Ӷ���֮��Ϊ3��4��
��2010?��������ģ��X��Y��Z��W��Ϊ����10���ӵ���������X��Y��ZΪ���ӣ�WΪ���ӣ���X��Z�����к��еĹ��õ��Ӷ���֮��Ϊ3��4��