��Ŀ����
��ͼ�У��ס��ҡ�����������ֱ��ʾ��ѧ��ѧ�г�����һ�ֵ��ʣ�������ĸ�ֱ��ʾһ�ֻ��������H��ˮ��Һ��dz��ɫ����Ӧ�ٵ�ԭ���ڹ�ҵ��������ұ��ҵ��������Ҫ��Ӧ�á�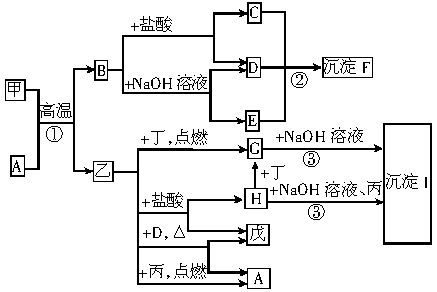
����д���пհף�
��1����ԭ�ӽṹʾ��ͼ��___________�����ĵ���ʽ��_______������I�Ļ�ѧʽ��________��
��2����Ӧ�ٵĻ�ѧ����ʽ��_________________����Ӧ�ڵ����ӷ���ʽ��____________________��
��3������D�ڼ���ʱ��Ӧ�Ļ�ѧ����ʽ��______
______________��������F���Ⱥ�õ��Ĺ������������������µ��Ļ�ѧ����ʽ��________________��
��1��
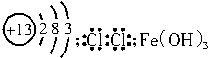
��2��3Fe3O4+8Al
 9Fe+4Al2O3��Al3++
9Fe+4Al2O3��Al3++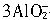 +6H2O=4Al��OH��3��
+6H2O=4Al��OH��3����3��3Fe+4H2O��g��
 Fe3O4+4H2����
Fe3O4+4H2����2Al2O3
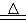
 4Al+3O2��
4Al+3O2�������������

��ϰ��ϵ�д�
 �����ҵ��ٿ���������������ϵ�д�
�����ҵ��ٿ���������������ϵ�д�
�����Ŀ






