��Ŀ����
2��1.52g ͭþ�Ͻ���ȫ�ܽ��� 50mL �ܶ�Ϊ 1.40g/mL����������Ϊ 63%��Ũ�����У��õ� NO2 �� N2 O4�Ļ������ 1120 mL����״��������Ӧ�����Һ�м��� 1.0 mol/L NaOH �� Һ������������ȫ������ʱ���õ� 2.54g ����������˵������ȷ���ǣ�������| A�� | �úϽ���ͭ��þ�����ʵ���֮���� 2��1 | |
| B�� | ��Ũ������ HNO3 �����ʵ���Ũ���� 14.0 mol/L | |
| C�� | NO2 �� N2 O4 �Ļ�������У�NO2 ����������� 80% | |
| D�� | �õ� 2.54 g ����ʱ������ NaOH ��Һ������� 600 mL |
���� A����������ȫ������ʱ���õ�2.54g����Ϊ������ͭ��������þ���ʳ�����������������Ϊ2.54g-1.52g=1.02g������n=$\frac{m}{M}$���������������ʵ��������ݵ���غ��֪�������ṩ�ĵ������ʵ������������������ʵ�������ͭ��þ�Ͻ���Cu��Mg�����ʵ����ֱ�Ϊxmol��ymol�������ṩ�ĵ������ʵ������������֮���з��̼���x��y��ֵ��
B������c=$\frac{1000�Ѧ�}{M}$����Ũ��������ʵ���Ũ�ȣ�
C������n=$\frac{V}{{V}_{m}}$����NO2��N2O4�����������ʵ�������������������ʵ���Ϊamol����ʾ�����������������ʵ��������ݵ���ת���غ��з��̼��㣻
D������n=cV�ɼ������������ʵ�������������ȫ������ʱ����Ӧ������Ϊ�����ƣ����ݵ�Ԫ���غ���������Ƶ����ʵ����������������غ��֪n��NaOH��=n��NaNO3�����ٸ���V=$\frac{n}{c}$������Ҫ����������Һ�������
��� �⣺A����������ȫ������ʱ���õ�2.54g����Ϊ������ͭ��������þ���ʳ�����������������Ϊ2.54g-1.52g=1.02g�������������ʵ���Ϊ$\frac{1.02g}{17g/mol}$=0.06mol�����ݵ���غ��֪�������ṩ�ĵ������ʵ������������������ʵ�������ͭ��þ�Ͻ���Cu��Mg�����ʵ����ֱ�Ϊxmol��ymol����2x+2y=0.06��64x+24y=1.52�����x=0.02��y=0.01���ʺϽ���ͭ��þ�����ʵ���֮���ǣ�2��1����A��ȷ��
B����Ũ�����ܶ�Ϊ1.40g/mL����������Ϊ63%�����Ũ��������ʵ���Ũ��Ϊ��$\frac{1000��1.4��63%}{63}$mol/L=14.0mol/L����B��ȷ��
C������£�NO2��N2O4�����������ʵ���Ϊ��$\frac{1.12L}{22.4L/mol}$=0.05mol����������������ʵ���Ϊamol�������������������ʵ���Ϊ��0.05-a��mol�����ݵ���ת���غ��֪��a��1+��0.05-a����2��1=0.06����ã�a=0.04��NO2���������Ϊ$\frac{0.04mol}{0.05mol}$��100%=80%����C��ȷ��
D����������ȫ������ʱ����Ӧ������Ϊ�����ƣ����ݵ�Ԫ���غ��֪�������Ƶ����ʵ���Ϊ0.05L��14mol/L-0.04mol-��0.05-0.04��mol��2=0.64mol�������������غ��֪n��NaOH��=n��NaNO3��=0.64mol������Ҫ����������Һ�����Ϊ��$\frac{0.64mol}{1mol/L}$=0.64L=640mL����D����
��ѡD��
���� ���⿼��������йؼ��㣬��Ŀ�Ѷ��еȣ����ⷴӦ�����Ĺ���Ϊ���ؼ����Ƕ�ѧ���ۺ������Ŀ��飬ע�������غ�˼���ڻ�ѧ�����е�Ӧ�÷�����

 �����ߴ���ϵ�д�
�����ߴ���ϵ�д�| A�� | X��Y�γɵĻ�������ܺ��зǼ��Լ� | |
| B�� | X�ĵ�����Z�ĵ����ڰ����ܾ��ҷ�Ӧ | |
| C�� | ��W��������ˮ�γɵ���Һ����������ˮ��ɱ������ | |
| D�� | ���Ӱ뾶��r��Z-����r��W3+�� |
| A�� | CO2 | B�� | SO2 | C�� | O2 | D�� | N2 |
 �������Ѽ��Ƚ��ȶ������� �˷�Ӧ�ɱ���ȩ����Ҳ���Ժϳ���ȩ���л������˵���в���ȷ���ǣ�������
�������Ѽ��Ƚ��ȶ������� �˷�Ӧ�ɱ���ȩ����Ҳ���Ժϳ���ȩ���л������˵���в���ȷ���ǣ�������| A�� | ���� 1mol ȩ��ʱҲ����2molCH3CH2OH | |
| B�� | �ϳ� 1mol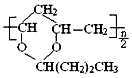 ��Ҫ 1mol ��Ҫ 1mol �� nmolCH3CH2CH2CHO �� nmolCH3CH2CH2CHO | |
| C�� | ͨ��������Ӧ�ϳɵ� ��˴Ź��������з����֮��Ϊ1��1��1��1��1��2��2��3 ��˴Ź��������з����֮��Ϊ1��1��1��1��1��2��2��3 | |
| D�� | 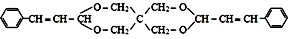 ���� C��CH2OH��4 �� ���� C��CH2OH��4 �� ͨ��������Ӧ�õ� ͨ��������Ӧ�õ� |
| A�� | NO2��ˮ��Ӧ��3NO2+H2O�T2NO3-+NO+2H+ | |
| B�� | ������������Һ�м����������SO32-+2H+�TSO2��+H2O | |
| C�� | ��ˮ����NO��NO2��NO+NO2+2OH-�T2NO2-+H2O | |
| D�� | NH4HCO3���ڹ���ŨNaOH��Һ�У�NH4++OH-�TNH3��+H2O |

 ��
��
 ��
��