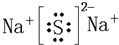��Ŀ����
�������ж�����Ԫ�����ʵ����ݣ�
�Իش��������⣺
��1������Ԫ��������ͬһ�������____________��Ԫ�آ���Ԫ�����ڱ��е�λ��Ϊ______________��
��2��Ԫ�آ١�����������ֻ����д�����бȽ��ȶ��Ļ�������ˮ��Ӧ�����ӷ���ʽ��____________��
��3��Ԫ�آ�ĵ��ʵĻ����ṹ��Ԫ����ͼ��ʾ������ʮ����ռ乹�ͣ�������20���ȱ������ε����һ����Ŀ�Ķ��ǣ�ÿ���������һ��ԭ�ӡ��˻����ṹ��Ԫ��_____��ԭ�ӹ��ɡ�

��1������ޡ�����ߣ���3���ڢ�A��
��2��2Na2O2+2H2O=4Na++4OH-+O2

��3��12
�����������

��ϰ��ϵ�д�
�����Ŀ