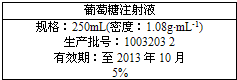
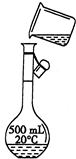
��Ŀ����
ʵ��������������480mL0.4mol?L-1��NaOH��Һ������������Һ�ľ�����̻ش��������⣺
��1�����Ƹ���ҺӦѡ��______mL����ƿ��
��2����������ƽ��ȡ______g����NaOH��
��3�����ƺõ�NaOH�������500mL�Ĵ��ձ��У���������ˮ���ò�������������ȫ�ܽ⣮����ȴ�����º��ձ��е���Һ�ò���������ת��������ƿ��
��4������������ˮϴ���ձ�2-3�Σ�����ÿ��ϴ�ӵ���Һ��ע��______������ζ�����ƿ��ʹ��Һ��;��ȣ�
��5��������ƿ�м�������ˮ��ֱ��Һ����̶���Լ1-2����ʱ����______�μ�����ˮ��Һ����̶������У��Ǻ�ƿ����ҡ�ȣ�
��6�����ƺõ���Һ______����ܡ����ܡ������ڴ��������ƿ�У�
��7���������ػ����ʵ����ƫ�͵���______��
A������ʱ�۲�Һ�����ӡ�B������ʱ�۲�Һ�温��
C��������NaOH��Һ�������ձ��� D������ƿ��ԭ������������ˮ��
��1�����Ƹ���ҺӦѡ��______mL����ƿ��
��2����������ƽ��ȡ______g����NaOH��
��3�����ƺõ�NaOH�������500mL�Ĵ��ձ��У���������ˮ���ò�������������ȫ�ܽ⣮����ȴ�����º��ձ��е���Һ�ò���������ת��������ƿ��
��4������������ˮϴ���ձ�2-3�Σ�����ÿ��ϴ�ӵ���Һ��ע��______������ζ�����ƿ��ʹ��Һ��;��ȣ�
��5��������ƿ�м�������ˮ��ֱ��Һ����̶���Լ1-2����ʱ����______�μ�����ˮ��Һ����̶������У��Ǻ�ƿ����ҡ�ȣ�
��6�����ƺõ���Һ______����ܡ����ܡ������ڴ��������ƿ�У�
��7���������ػ����ʵ����ƫ�͵���______��
A������ʱ�۲�Һ�����ӡ�B������ʱ�۲�Һ�温��
C��������NaOH��Һ�������ձ��� D������ƿ��ԭ������������ˮ��
��1��ʵ������480ml����ƿ��ֻ������500ml��Һ���ʴ�Ϊ��500��
��2������500mL 0.4mol?L-1��NaOH��Һ�����ʵ����ʵ���Ϊ0.4mol/L��0.5L=0.2mol�����ʵ�����Ϊ��0.2mol��40g/mol=8.0g���ʴ�Ϊ��8.0��
��4����5�����������м��㡢�������ܽ⡢��Һ��ϴ����Һ�����ݡ�ҡ�ȵȲ�����һ����������ƽ��������ҩ��ȡ��ҩƷ�����ձ����ܽ⣨������Ͳ��ȡˮ�����ձ��������ò��������裬�����ܽ⣬��ȴ��ת�Ƶ�500mL����ƿ�У����ò�����������ϴ���ձ���������2-3�Σ�����ϴ��Һ��������ƿ�У���ˮ��Һ�����̶���1��2cmʱ�����ý�ͷ�ιܵμӣ�����ݵߵ�ҡ�ȣ�
�ʴ�Ϊ��500mL����ƿ����ͷ�ιܣ�
��6�����ƺõ���Һ���ܳ��ڴ��������ƿ�У��ʴ�Ϊ�����ܣ�
��7��A������ʱ�۲�Һ�����ӣ�����������Һ�����ƫ��������Һ��Ũ��ƫ�ͣ���A��ȷ����
B������ʱ�۲�Һ�温�ӣ�����������Һ�����ƫС��������Һ��Ũ��ƫ�ߣ���B����
C��������NaOH��Һ�������ձ��У��������ʵ�����ƫС��������Һ��Ũ��ƫ�ͣ���C��ȷ��
D������ƿ��ԭ������������ˮ������Һ�������Ӱ�죬������Һ��Ũ�Ȳ��䣬��D����
��ѡAC��
��2������500mL 0.4mol?L-1��NaOH��Һ�����ʵ����ʵ���Ϊ0.4mol/L��0.5L=0.2mol�����ʵ�����Ϊ��0.2mol��40g/mol=8.0g���ʴ�Ϊ��8.0��
��4����5�����������м��㡢�������ܽ⡢��Һ��ϴ����Һ�����ݡ�ҡ�ȵȲ�����һ����������ƽ��������ҩ��ȡ��ҩƷ�����ձ����ܽ⣨������Ͳ��ȡˮ�����ձ��������ò��������裬�����ܽ⣬��ȴ��ת�Ƶ�500mL����ƿ�У����ò�����������ϴ���ձ���������2-3�Σ�����ϴ��Һ��������ƿ�У���ˮ��Һ�����̶���1��2cmʱ�����ý�ͷ�ιܵμӣ�����ݵߵ�ҡ�ȣ�
�ʴ�Ϊ��500mL����ƿ����ͷ�ιܣ�
��6�����ƺõ���Һ���ܳ��ڴ��������ƿ�У��ʴ�Ϊ�����ܣ�
��7��A������ʱ�۲�Һ�����ӣ�����������Һ�����ƫ��������Һ��Ũ��ƫ�ͣ���A��ȷ����
B������ʱ�۲�Һ�温�ӣ�����������Һ�����ƫС��������Һ��Ũ��ƫ�ߣ���B����
C��������NaOH��Һ�������ձ��У��������ʵ�����ƫС��������Һ��Ũ��ƫ�ͣ���C��ȷ��
D������ƿ��ԭ������������ˮ������Һ�������Ӱ�죬������Һ��Ũ�Ȳ��䣬��D����
��ѡAC��

��ϰ��ϵ�д�
�����Ŀ