��Ŀ����
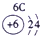
��13�֣��±�ΪԪ�����ڱ���һ���֣�A��B��D��E��G��L��QΪ����Ԫ�ء�����L�ĵ��������������������������ʮ����Ҫ�����ã�Q�Ļ���������к�ɫ����ɫ����ɫ����ɫ�ȶ�����ɫ��

(1)��ش��������⣺
��д��G���Ľṹʾ��ͼ��________��
��Ԫ��L�����ڱ��е�λ�ã�________��
��Ԫ��Q�ĵ���Ϊ��ɫ��д������Q�ڿ���������Ļ�ѧ����ʽ��_______________��
(2)��A��D��E��G��L����Ԫ����ɵ�����[DL(EA)nG6��n]m(����m��nΪ��������n<5��m<10)�㷺Ӧ�����ճ�������ˮ��ҵ��ˮ�Ĵ�����ij�������Թ�ҵ��������(D��L�ĵ��ʼ���������)Ϊԭ����ȡ[DL(EA)nG6��n]m����Ƶ������������£�

��ѡ�õ��Լ�XӦΪ________��
�����й���[DL(EA)nG6��n]m��˵����ȷ����________(�����)��
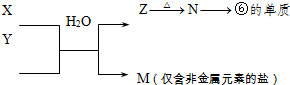
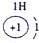
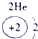
A����������LԪ�صĻ��ϼ�Ϊ��2
B��������������̬������
C����������ǿ���ǿ������Һ�о������ȶ�����
D������������ˮ�ܵ����(EA)���ӣ���һ�ּ�
��д���������̵�������Һ�м���BGE3ʱ��������Ӧ�����ӷ���ʽ��_______________��
��1���� �ڵ������ڵ� ���� ��2Cu��O2��CO2��H2O===Cu2(OH)2CO3
�ڵ������ڵ� ���� ��2Cu��O2��CO2��H2O===Cu2(OH)2CO3
(2)�ٹ������ᡡ��C����6Fe2����ClO��6H��===6Fe3����Cl����3H2O
����������1���������ڱ��Ľṹ��Ԫ�������ɵ�Ӧ�á�G��17��Ԫ����Ԫ�ء�L����Ԫ�أ�Q��ͭԪ�أ��ڿ����б����������γ�ͭ�̣�����ʽΪ
Cu��O2��CO2��H2O===Cu2(OH)2CO3��
��2��A��D��E��G��L����Ԫ�طֱ���H��Al��O��Cl��Fe���ڻ�����[AlFe(OH)nCl6��n]m�и������һ����6�����������Ļ��ϼ۾��ǣ�3�ۡ�����Һ�иû�����ɵ����Fe3����Al3����Cl����OH��������ֻ��ѡ��C��ȷ����Ϊ�����������ʣ������ܽ�ʱ�����ᡣ������ܽ�ʱ�����������ӣ���BGE3��NaClO3���������ԣ��ɽ�Fe2����������Fe3��������ʽΪ6Fe2����ClO��6H��===6Fe3����Cl����3H2O��









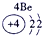
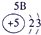
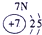
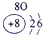
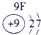
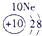
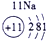





 ��ʾ����
��ʾ����