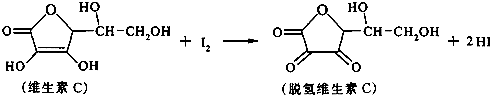��Ŀ����
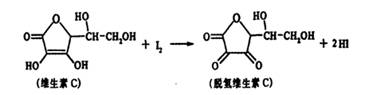
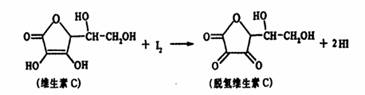
ά����C������ʽΪC6H8O6�����н�ǿ�Ļ�ԭ�ԣ������ڿ������ױ��������京����ͨ������������Һ������֪��Һ��I2��Һ���еζ����÷�Ӧ�Ļ�ѧ����ʽ���£�
C6H8O6+I2![]() C6H6O6+2HI
C6H6O6+2HI
�����ⶨij��Ʒ��ά����C�ĺ���������IJ��輰��õ��������¡�ȡ10mL 6mol��L��1CH3COOH������100mL����ˮ������Һ������к������ȴ����ȷ��ȡ0��2000g��Ʒ���ܽ���������ȴ����Һ�У�����1mL����ָʾ����������Ũ��Ϊ0��05000mol��L��1��I2��Һ���еζ���ֱ����Һ�е���ɫ��������Ϊֹ��������21��00mLI2��Һ��
��1��Ϊ�μ����CH3COOHϡ��ҺҪ�Ⱦ���С���ȴ�����ʹ�ã�
��2��������Ʒ��ά����C������������
��1�������Ϊ�˳�ȥ��Һ����Һ��O2������ά����C��O2��������ȴ��Ϊ�˼����ζ�������ά����C��Һ���Ͽ����Ӵ�ʱ���������ٶȡ�
��2��![]() =92��40%
=92��40%

��ϰ��ϵ�д�
 ��Ǭ����������ҵ���ּ����ӱ����������ϵ�д�
��Ǭ����������ҵ���ּ����ӱ����������ϵ�д�
�����Ŀ