��Ŀ����
�ش����и�С�⣺
��1����֪ ��ˮ�еĵ��뷽��ʽΪ
��ˮ�еĵ��뷽��ʽΪ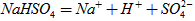 ����
����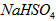 ��Һ
��Һ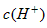
__________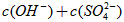 ���>����=����<������
���>����=����<������
��1����֪
 ��ˮ�еĵ��뷽��ʽΪ
��ˮ�еĵ��뷽��ʽΪ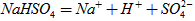 ����
����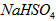 ��Һ
��Һ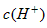
__________
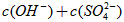 ���>����=����<������
���>����=����<������ 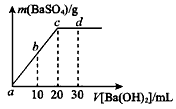
��2�������£���1.0mol/L��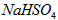 ��Һ����μ�������ʵ���Ũ�ȵ�
��Һ����μ�������ʵ���Ũ�ȵ�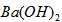 ��Һ�����������������������������Һ�������ϵ��ͼ��ʾ��a��b��c��d�ֱ��ʾʵ��ʱ��ͬ�ε���Һ������b���ʾ��Һ��_________������ԡ������ԡ����ԡ�����c����ʾ�����ӷ���ʽΪ___________________��
��Һ�����������������������������Һ�������ϵ��ͼ��ʾ��a��b��c��d�ֱ��ʾʵ��ʱ��ͬ�ε���Һ������b���ʾ��Һ��_________������ԡ������ԡ����ԡ�����c����ʾ�����ӷ���ʽΪ___________________��
��3����T��ʱ����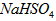 ������뵽pH=6������ˮ�У������¶Ȳ��䣬�����Һ��pHΪ2��T�潫
������뵽pH=6������ˮ�У������¶Ȳ��䣬�����Һ��pHΪ2��T�潫
__________25�棨����ڡ����ڡ�����KwΪ________���ڸ���Һ����ˮ������� Ϊ
Ϊ
_________mol��L-1��T��ʱ����pH=11��NaOH��ҺV1L��pH=1��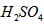 ��ҺV2L��ϣ����Ϻ���Һ�����Ϊԭ����Һ���֮�ͣ������û����Һ��pH=2����V1: V2 =____________������Һ�и������ӵ�Ũ���ɴ�С������˳��Ϊ_________________________��
��ҺV2L��ϣ����Ϻ���Һ�����Ϊԭ����Һ���֮�ͣ������û����Һ��pH=2����V1: V2 =____________������Һ�и������ӵ�Ũ���ɴ�С������˳��Ϊ_________________________��
��4��0.1 mol��L-1pHΪ4��NaHB��Һ��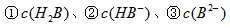 �ɴ�С��˳��Ϊ
�ɴ�С��˳��Ϊ
_______________��
��5����0.1 mol��L-1�Ģ� ��Һ����
��Һ����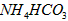 ��Һ����
��Һ���� ��Һ�У�
��Һ�У�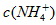 �ɴ�С������˳��Ϊ_____________________��
�ɴ�С������˳��Ϊ_____________________��
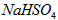 ��Һ����μ�������ʵ���Ũ�ȵ�
��Һ����μ�������ʵ���Ũ�ȵ�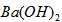 ��Һ�����������������������������Һ�������ϵ��ͼ��ʾ��a��b��c��d�ֱ��ʾʵ��ʱ��ͬ�ε���Һ������b���ʾ��Һ��_________������ԡ������ԡ����ԡ�����c����ʾ�����ӷ���ʽΪ___________________��
��Һ�����������������������������Һ�������ϵ��ͼ��ʾ��a��b��c��d�ֱ��ʾʵ��ʱ��ͬ�ε���Һ������b���ʾ��Һ��_________������ԡ������ԡ����ԡ�����c����ʾ�����ӷ���ʽΪ___________________����3����T��ʱ����
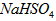 ������뵽pH=6������ˮ�У������¶Ȳ��䣬�����Һ��pHΪ2��T�潫
������뵽pH=6������ˮ�У������¶Ȳ��䣬�����Һ��pHΪ2��T�潫__________25�棨����ڡ����ڡ�����KwΪ________���ڸ���Һ����ˮ�������
 Ϊ
Ϊ_________mol��L-1��T��ʱ����pH=11��NaOH��ҺV1L��pH=1��
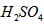 ��ҺV2L��ϣ����Ϻ���Һ�����Ϊԭ����Һ���֮�ͣ������û����Һ��pH=2����V1: V2 =____________������Һ�и������ӵ�Ũ���ɴ�С������˳��Ϊ_________________________��
��ҺV2L��ϣ����Ϻ���Һ�����Ϊԭ����Һ���֮�ͣ������û����Һ��pH=2����V1: V2 =____________������Һ�и������ӵ�Ũ���ɴ�С������˳��Ϊ_________________________����4��0.1 mol��L-1pHΪ4��NaHB��Һ��
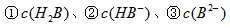 �ɴ�С��˳��Ϊ
�ɴ�С��˳��Ϊ_______________��
��5����0.1 mol��L-1�Ģ�
 ��Һ����
��Һ����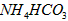 ��Һ����
��Һ���� ��Һ�У�
��Һ�У�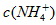 �ɴ�С������˳��Ϊ_____________________��
�ɴ�С������˳��Ϊ_____________________�� ��1��=
��2�����ԣ�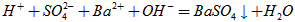
��3�����ڣ�10-12��10-10��9:11��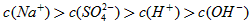
��4����>��>��
��5����>��>��>��
��2�����ԣ�
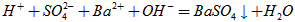
��3�����ڣ�10-12��10-10��9:11��
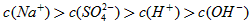
��4����>��>��
��5����>��>��>��

��ϰ��ϵ�д�
�����Ŀ
 �ش����и�С�⣺
�ش����и�С�⣺