��Ŀ����
�����Ǽ�����Ҫ�Ļ���ԭ�ϣ���ҵ�Ͻ�������NO2���ܱ���������ˮ��η���ѭ�������Ʊ����ᡣ
��1����ҵ����ˮ����NO2����HNO3�����ɵ����徭������������յ�ѭ���������ת��Ϊ����(�ٶ�����������������ʧ)����д��������Ӧ�Ļ�ѧ����ʽ��
______________________________________________________________��
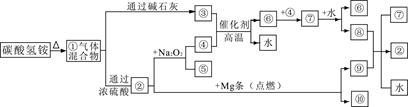
��2��Ϊ��֤��NOҲ������������ˮ��ͬ��Ӧ����HNO3��ijѧ���������ͼ��ʾװ��(�йؼг�װ������ȥ)��
�ټ��װ�����������ú�Ϊ�۲쵽NO�������ɣ���K1���ر�K2��Ӧ��U�ιܵij��ܿ�ע��ϡ����������������Ѹ�ٹر�K1���۲쵽U�ι��ڵ�������____________________________________________________��
��װ�â��з�����Ӧ�Ļ�ѧ����ʽΪ_______________________________��
��պNaOH��Һ�����ŵ�������___________________________________��
�ܴ�K2����װ�â��г��������е��������ɫ��K3����Ӧһ��ʱ����������в�δ����Һ�塣��Ƽ������鳤�������е������Ƿ�NO__________________________________________________��
��1��4NO2��O2��2H2O=4HNO3(��ֳ���������ʽд)
��2����U�ι��Ҳེ�����ء�U�ι����Һ������Ҷˣ�ͭ˿�����ܽ⣬������ɫ���壬��Һ����(���ٴ�����)����2H2O2 2H2O��O2��
2H2O��O2��
�����յ����������ֹ��Ⱦ����
���ٴ�K3�����۲쵽��������������Ѹ�ٱ�Ϊ����ɫ����֤��������NO��������ɫ�仯����֤������NO(����������Ҳ��)
�����������������ʵ��Ŀ�ĺ������õ��Լ������ƶϣ���װ�õ��������Ʊ�NO����װ�õ��������Ʊ�O2����װ�õ�������֤��NOҲ������������ˮ��ͬ��������HNO3��
��1��NO2�������������ˮ���յ�ѭ�����������ת��Ϊ����Ļ�ѧ����ʽΪ4NO2��O2��2H2O=4HNO3��
��2����ע��ϡ����Ӧ��U�ι��Ҳེ�����أ�Ѹ�ٹر�K1��U�ι����Һ������Ҷˣ�ͭ˿�����ܽ⣬������ɫ���壬��Һ������
��װ�â��з�����Ӧ�Ļ�ѧ����ʽΪ��2H2O2 2H2O��O2����
2H2O��O2����
��պNaOH��Һ�����ŵ����������յ����������ֹ��Ⱦ������
���ٴ�K3�����۲쵽��������������Ѹ�ٱ�Ϊ����ɫ����֤��������NO��������ɫ�仯����֤������NO��
���㣺���⿼��ʵ��װ�ú�ʵ�鷽���ķ�������Ӧ�������������ѧ����ʽ����д��

��ѧ������ũҵ�����о�����Ҫ�����ã���ϵ��ũҵ�����Ƿ���ա���ѧ������ũҵ�����еĹ㷺Ӧ�ã�Ϊ��ѧ���ϵĴ��ģ��ҵ�����ṩ����̨��
��1���ںϳɰ����豸(�ϳ���)�У������Ƚ�������Ŀ����______________��
��2����������Ĺ����г������һЩ���������һ��ɲ����������ַ���������
��Һ���շ���NO��NO2��2NaOH=2NaNO2��H2O
����ԭ����8NH3��6NO2 7N2��12H2O(NOҲ�����Ƶķ�Ӧ)
7N2��12H2O(NOҲ�����Ƶķ�Ӧ)
�����ɫ��ѧ�ĽǶȷ������ַ��������ӣ�____________________��
��3��ij���ʳ���NH3�Ʊ�NH4NO3����֪����NH3��NO�IJ�����96%��NO��HNO3�IJ�����92%������HNO3����ȥ��NH3������ռ�ܺ���NH3����(�������������)��________%��
��4���������һ�ֳ��õĵ��ʣ��������ʹ�øû���ʱ����Ӧע���������������±�(������Ӧע���������ɼ���)��
| | ע������ | ���� |
| �� | ______________ | ____________ |
| �� | ________________ | ____________ |
Na2SO3�dz��õĿ�������
��1��ʵ����ͨ����Ũ���ᣨ1��1����Na2SO3���Ʊ�SO2���壬
��Ӧ����ʽΪ�� ���Ʊ���SO2������ͨ������ˮ���������и�����ܸ���SO2������ǣ� ��
A.Ũ���� B.��ʯ�� C.��ˮCaCl2
��2�� ����SO2����ͨ��NaOH��Һ�пɵ�NaOH��Na2SO3�Ļ����Һ����û����Һ�м���������ˮ������Һ��Ϊ��ɫ��������Һ��Br2��Na2SO3����������ԭ��Ӧ����Ӧ�����ӷ���ʽΪ______________��
��3����Ӧ�����Һ����SO32����SO42����Br����OH���������ӣ�����д��������SO32����SO42����Br����ʵ�鱨�棻
��ѡ�Լ���2 mol��L��1HCl��1 mol��L��1H2SO4��1mol��L��1HNO3��1 mol��L��1BaCl2��
1 mol��L��1Ba(NO3)2��0.1 mol��L��1AgNO3��CCl4���������Ʊ�����ˮ�����Ʊ�����ˮ��
| ��� | ʵ����� | Ԥ������ͽ��� |
| ����� | ȡ��������Һ���Թ�A�У��μ�2 mol��L��1HCl����Һ�����ԣ����뼸��________(���Լ�)���� | ________��֤������Һ�к�SO32- |
| ����� | ��ȡ��������Һ���Թ�B�У����� ���ٵμ����� 1 mol��L��1 BaCl2��Һ | |
| ����� | ��ȡ��������Һ���Թ�C�У� �������ú�۲���ɫ | ��Һ�ֲ㣬�ϲ�Һ��ʳȺ�ɫ��֤������Һ�к�Br- |
��������;ʮ�ֹ㷺�������ھ�ˮ�����������⣬�������������ᡢ��ȡ�
��1����ҵ�Ͽ���������ʯ����Ϊԭ������Ư�ۣ�Ư�۵���Ч�ɷ��� ��
��2����ҵ�������뵼����Ϲ���������£�
��д���Ʊ��ֹ�ʱ��Ӧ�Ļ�ѧ����ʽ�� ��
�ڴֹ���������Ӧ��õ��е�ϵ͵�Һ̬���Ȼ����г�����һЩ�߷е㡢�ѻӷ���Һ̬���ʣ�������з����ᴿ�����ᴿ����Ϊ ������ĸ����
| A������ | B������ | C����ȡ | D���ᾧ |