��Ŀ����
12�����г�����pH=2�������pH=2�Ĵ�����Һ�ң���������в����ش����⣺��1��������0.1mol/L��CH3COOH��Һ��ˮϡ���̣����б���ʽ������һ����С����A��
A��c��H+�� B��$\frac{c��{H}^{+}��}{c��C{H}_{3}COOH��}$ C��c��H+��•c��OH-�� D��$\frac{c��O{H}^{-}��}{c��{H}^{+}��}$
��2����ͬ�����£�ȡ������ļס�������Һ����ϡ��100����ϡ�ͺ����Һ����pH��С��ϵΪ��pH���ף���pH���ң��� ���������������=������
��3����֪25��ʱ����ĵ���ƽ�ⳣ�����£�
| ��ѧʽ | CH3COOH | H2CO3 | HClO |
| ����ƽ�ⳣ��K1 | 1.8��10-5 | 4.3��10-7 | 3.0��10-8 |
| K2 | -- | 5.6��10-11 | -- |
CH3COOH+Na2CO3����������2CH3COOH+CO32-=H2O+2CH3COO-+CO2����
HClO+Na2CO3����������HClO+CO32-=ClO-+HCO3-��
���� ��1��CH3COOH��Һ��ˮϡ���̣��ٽ����룬c��H+����С��c��OH-������Kw���䣻
��2���Ȼ�����ǿ����ʣ���ȫ���룬������������ʣ���Һ�д��ڵ���ƽ�⣬��ˮϡ�ʹٽ�������룻
��3�����볣��Խ������Խǿ������ǿ��������ԭ��������
��� �⣺��1��A��CH3COOH��Һ��ˮϡ���̣��ٽ����룬c��H+����С����Aѡ��
B��$\frac{c��{H}^{+}��}{c��C{H}_{3}COOH��}$=$\frac{n��{H}^{+}��}{n��C{H}_{3}COOH��}$����ϡ�����б�ֵ���B��ѡ��
C��ϡ���̣��ٽ����룬c��H+����С��c��OH-������c��H+��•c��OH-��=Kw��Kw���䣬��C��ѡ��
D��ϡ���̣��ٽ����룬c��H+����С��c��OH-��������$\frac{c��O{H}^{-}��}{c��{H}^{+}��}$���D��ѡ��
�ʴ�Ϊ��A��
��2���Ȼ�����ǿ����ʣ���ȫ���룬������������ʣ���Һ�д��ڵ���ƽ�⣬��ˮϡ�ʹٽ�������룬���Դ�����Һ��������Ũ�ȼ�С�̶ȴ���������Һ��������Ũ�ȼ�С�̶ȣ����������pH���ڴ��ᣬ
�ʴ�Ϊ������
��3�����볣��Խ������Խǿ�����ԣ�CH3COOH��H2CO3��HClO��HCO3-��
CH3COOH������Na2CO3��Һ��Ӧ���ɴ����ƺͶ�����̼����Ӧ�ķ���ʽΪ��2CH3COOH+CO32-=H2O+2CH3COO-+CO2����
HClO��������Na2CO3��Һ��Ӧ����NaClO��NaHCO3����Ӧ�ķ���ʽΪ��HClO+CO32-=ClO-+HCO3-��
�ʴ�Ϊ��2CH3COOH+CO32-=H2O+2CH3COO-+CO2����HClO+CO32-=ClO-+HCO3-��
���� ���⿼����������ʵĵ��룬��ȷ������ʵ����ص��ǽⱾ��ؼ���ע����ͬŨ�ȵIJ�ͬ��ϡ��ʱ��pH�仯��С���Ѷ��еȣ�

 ��Ԫȫ��������ϵ�д�
��Ԫȫ��������ϵ�д� �»ƸԱ����ܾ�ϵ�д�
�»ƸԱ����ܾ�ϵ�д�| A�� | �õ���ʽ��ʾHCl���γɹ��̣�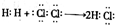 | |
| B�� | Mg2+�Ľṹʾ��ͼ��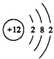 | |
| C�� | HClO�Ľṹʽ��H-Cl-O | |
| D�� | ������Ϊ133��������Ϊ78���ԭ�ӣ�${\;}_{55}^{133}$Cs |
| A�� | �ڢݢ� | B�� | �ڢۢݢ� | C�� | �ڢۢ� | D�� | �٢ۢ� |
| A�� | Ca��HCO3��2��Һ�����NaOH��Һ��Ӧ���Ƶ�Ca��OH��2 | |
| B�� | ��CO2���Լ���NaAlO2��Һ��CH3COONa��Һ | |
| C�� | ϡ���������ᡢ�Ȼ�����Һ��Ϊ���� | |
| D�� | ��BaCl2������Һ����AgNO3��Һ��K2SO4��Һ |
| A�� | 28g���������е�ԭ����ĿΪNA | |
| B�� | ���³�ѹ�£�32gO2���е�Oԭ����ΪNA | |
| C�� | 1mol�����������NA��������ӵ�����֮����� | |
| D�� | ��״���£�22.4L �������Ȳ�Ļ�����������ķ�����ΪNA |


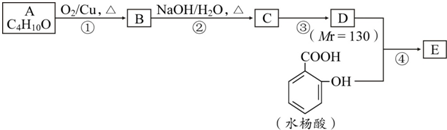

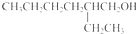 ��
��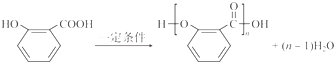 ��
��
 ���������ḻ��ʣ�����Ľṹ�����ӣ��˽̰�γ̱����н̲ġ�ѡ��3��������չʾ��һЩ���;���Ľṹ�����ʵ����֪ʶ���磺�ɱ����������ʯ��ʯī���ǡ��ء�þ��ͭ��NaCl��CsCl��CaF2����ͼ��һ����������ͼ�������������������ʷ�Χ�ڻش��������⣺
���������ḻ��ʣ�����Ľṹ�����ӣ��˽̰�γ̱����н̲ġ�ѡ��3��������չʾ��һЩ���;���Ľṹ�����ʵ����֪ʶ���磺�ɱ����������ʯ��ʯī���ǡ��ء�þ��ͭ��NaCl��CsCl��CaF2����ͼ��һ����������ͼ�������������������ʷ�Χ�ڻش��������⣺