��Ŀ����
3����֪A����Է�������Ϊ92.5�ı���һ�ȴ����������к�����������C�Ǹ߷��ӻ����ת����ϵ��ͼ��ʾ���ش��������⣺
��1��A�ķ���ʽΪC4H9Cl��F�й����ŵ�����Ϊȩ�����ǻ���
��2��д���������ʵĽṹ��ʽ��E��CH3��2C��OH��-CH2OH����Ũ������������£�����������Ԫ��״�������ṹ��ʽΪ
 ���÷��ӵĺ˴Ź���������2�ַ壮
���÷��ӵĺ˴Ź���������2�ַ壮��3��д�����з�Ӧ�Ļ�ѧ����ʽ��G��һ�����������ɸ߷��ӻ����
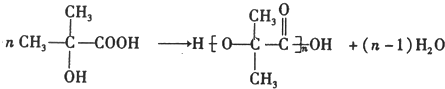 ��F��G�ķ�Ӧ����CH3��2C��OH��-CHO+2Cu��OH��2$\stackrel{��}{��}$��CH3��2C��OH��-COOH+Cu2O��+2H2O��
��F��G�ķ�Ӧ����CH3��2C��OH��-CHO+2Cu��OH��2$\stackrel{��}{��}$��CH3��2C��OH��-COOH+Cu2O��+2H2O����4����������������G��ͬ���칹����5�֣�
���������� ���ܷ���������Ӧ ����������Ʒ�Ӧ�ų����壮
���� A����Է�������Ϊ92.5�ı���һ�ȴ����������к���1��Clԭ�ӣ�ʣ������ʽ��Ϊ92.5-35.5=57���������෨$\frac{57}{12}$=4��9��������Ϊ-C4H9��A�ķ���ʽΪC4H9Cl�������к�������������AΪC��CH3��3Cl��A���������ƴ���Һ�з�����ȥ��Ӧ�õ�BΪ��CH3��2C=CH2��B�����Ӿ۷�Ӧת��Ϊ�߷��ӻ�����CΪ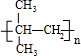 ����CH3��2C=CH2���巢���ӳɷ�Ӧ����DΪ��CH3��2CBr-CH2Br��D�ڼ���������ˮ��ת��ΪEΪ��CH3��2C��OH��-CH2OH��E����������Ӧ����FΪ��CH3��2C��OH��-CHO��F��������õ�G����GΪ��CH3��2C��OH��-COOH���ݴ˽��
����CH3��2C=CH2���巢���ӳɷ�Ӧ����DΪ��CH3��2CBr-CH2Br��D�ڼ���������ˮ��ת��ΪEΪ��CH3��2C��OH��-CH2OH��E����������Ӧ����FΪ��CH3��2C��OH��-CHO��F��������õ�G����GΪ��CH3��2C��OH��-COOH���ݴ˽��
��� �⣺A����Է�������Ϊ92.5�ı���һ�ȴ����������к���1��Clԭ�ӣ�ʣ������ʽ��Ϊ92.5-35.5=57���������෨$\frac{57}{12}$=4��9��������Ϊ-C4H9��A�ķ���ʽΪC4H9Cl�������к�������������AΪC��CH3��3Cl��A���������ƴ���Һ�з�����ȥ��Ӧ�õ�BΪ��CH3��2C=CH2��B�����Ӿ۷�Ӧת��Ϊ�߷��ӻ�����CΪ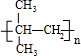 ����CH3��2C=CH2���巢���ӳɷ�Ӧ����DΪ��CH3��2CBr-CH2Br��D�ڼ���������ˮ��ת��ΪEΪ��CH3��2C��OH��-CH2OH��E����������Ӧ����FΪ��CH3��2C��OH��-CHO��F��������õ�G����GΪ��CH3��2C��OH��-COOH��
����CH3��2C=CH2���巢���ӳɷ�Ӧ����DΪ��CH3��2CBr-CH2Br��D�ڼ���������ˮ��ת��ΪEΪ��CH3��2C��OH��-CH2OH��E����������Ӧ����FΪ��CH3��2C��OH��-CHO��F��������õ�G����GΪ��CH3��2C��OH��-COOH��
��1����������ķ�����֪��A�ķ���ʽΪC4H9Cl��FΪ��CH3��2C��OH��-CHO��F�й����ŵ�����Ϊȩ�����ǻ���
�ʴ�Ϊ��C4H9Cl��ȩ�����ǻ���
��2��EΪ��CH3��2C��OH��-CH2OH����Ũ������������£�����������Ԫ��״�������ṹ��ʽΪ  ���÷��ӵĺ˴Ź���������2�ַ壬
���÷��ӵĺ˴Ź���������2�ַ壬
�ʴ�Ϊ����CH3��2C��OH��-CH2OH�� ��2��
��2��
��3��GΪ��CH3��2C��OH��-COOH��G��һ�����������ɸ߷��ӻ������Ӧ�Ļ�ѧ����ʽΪ��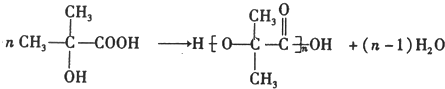 ��F��G�ķ�Ӧ�Ļ�ѧ����ʽΪ��CH3��2C��OH��-CHO+2Cu��OH��2$\stackrel{��}{��}$��CH3��2C��OH��-COOH+Cu2O��+2H2O��
��F��G�ķ�Ӧ�Ļ�ѧ����ʽΪ��CH3��2C��OH��-CHO+2Cu��OH��2$\stackrel{��}{��}$��CH3��2C��OH��-COOH+Cu2O��+2H2O��
�ʴ�Ϊ��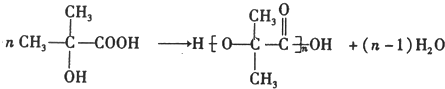 ����CH3��2C��OH��-CHO+2Cu��OH��2$\stackrel{��}{��}$��CH3��2C��OH��-COOH+Cu2O��+2H2O��
����CH3��2C��OH��-CHO+2Cu��OH��2$\stackrel{��}{��}$��CH3��2C��OH��-COOH+Cu2O��+2H2O��
��4��GΪ��CH3��2C��OH��-COOH��G��ͬ���칹�������࣬�ܷ���������Ӧ��Ϊ����������������Ʒ�Ӧ�ų����壬������-OH������������ͬ���칹���У�HOCH2CH2CH2OOCH��CH3CH��OH��CH2OOCH��CH3CH2CH��OH��OOCH��HCOOC��CH3��CH2OH��HCOOC��OH����CH3��2�������֣�
�ʴ�Ϊ��5��
���� ���⿼���л�����ƶϣ��Ƕ��л���ѧ֪ʶ���ۺϿ��飬�ƶ�A�Ľṹ�ǽ���Ĺؼ����ܽϺõĿ���ѧ���ķ�����˼ά�������Ѷ��еȣ�

| A�� | ���¶Ȳ��䣬�����������һ������ʱA��Ũ����ԭ����0.48������a+b����d | |
| B�� | ��������Ӧ��ʼƽ��ʱA��B��ת������ȣ���A��B�����ʵ���֮��Ϊa��b | |
| C�� | ��ƽ����ϵ������M mol���ٳ���bmolB���ﵽƽ��������Ϊ��M+b��mol����a+b=c+d | |
| D�� | ��a+b=d�����������¶�����ʱ��ƽ�������ƶ��������������ѹǿ���� |

| A�� | ͼ�����еķ�Ӧ��Ϊ������ԭ��Ӧ | |
| B�� | ��Ӧ�٢ھ�Ϊ���ӷ�Ӧ | |
| C�� | ��Ӧ�ڢ۶��ܲ����������Ҳ���������������Al��������ת�Ƶ������ֱ���� | |
| D�� | ��Ӧ�٢ڢ�����������ԭ�� |
��1��ȡ��Ƭά����C��ѹ�������100mLˮ�й��ˣ�ȡ������Һ�����еμӼ�����ɫʯ����Һ����Һ��죬˵��ά����C��ˮ��Һ�����ԣ�
��2��ά����C�����Ի����бȽ��ȶ����ڸ���ʱ�ױ��ƻ���������һ�룬���ʳ�ûƹϣ����ܳ�����ûƹ��зḻ��ά����C��ʳ�ûƹ�ʱ��üӵ�����裮
��ij����ʳƷ�����װ��ǩ�ϵIJ����������£�
| ��Ҫԭ�ϣ�̼��� ʳ�÷�����ÿ��һ�Σ�ÿ��һƬ����ʳ�� |
��2�����úñ���ʳƷ�е�̼�����θҺ�е����ᷢ����Ӧ�Ļ�ѧ����ʽ��CaCO3+2HCl=CaCl2+H2O+CO2����
��3��ʳ�÷����н�ʳ������������Ӧ��ĽӴ�������ٽ��Ƶ����գ�
 ���ΪԪ�����ڱ���һ���֣������Ԫ�آ١����ڱ��е�λ�ã��ش��������⣺
���ΪԪ�����ڱ���һ���֣������Ԫ�آ١����ڱ��е�λ�ã��ش��������⣺ ��
��