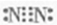��Ŀ����
��ͼ��ʾ��

����֪�ס��ҡ���������Ϊǰ������Ԫ�صĵ��ʡ�
�� ��һ���������¼�����ͼ��붡�������ʵ���֮��1��3 ��Ӧ���ֱ�����X��Y���ڲ�����Ԫ�ؼ׳ʸ��ۡ�
����һ������������������붡�������ʵ���֮��1��2��Ӧ���ֱ�����Z��W���ڲ�����Ԫ���ҳʸ��ۡ� ��գ�
��1������___________������____________
��2���������Ӧ����X�Ļ�ѧ����ʽ______________________________
��3�����붡��Ӧ����W�Ļ�ѧ����ʽΪ___________________________
�� ��һ���������¼�����ͼ��붡�������ʵ���֮��1��3 ��Ӧ���ֱ�����X��Y���ڲ�����Ԫ�ؼ׳ʸ��ۡ�
����һ������������������붡�������ʵ���֮��1��2��Ӧ���ֱ�����Z��W���ڲ�����Ԫ���ҳʸ��ۡ� ��գ�
��1������___________������____________
��2���������Ӧ����X�Ļ�ѧ����ʽ______________________________
��3�����붡��Ӧ����W�Ļ�ѧ����ʽΪ___________________________
(1)N2 O2
(2)N2+3Mg Mg3N2 (
Mg3N2 ( )
)
(3)O2+2H2 2H2O (O2+2Mg
2H2O (O2+2Mg 2MgO)
2MgO)
(2)N2+3Mg
 Mg3N2 (
Mg3N2 ( )
)(3)O2+2H2
 2H2O (O2+2Mg
2H2O (O2+2Mg 2MgO)
2MgO) 
��ϰ��ϵ�д�
�����Ŀ
 ��2009?���գ����������������Ե�10-�ǻ�ϲ����Ľṹ��ͼ��ʾ�����й���10-�ǻ�ϲ�����˵����ȷ���ǣ�������
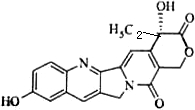
��2009?���գ����������������Ե�10-�ǻ�ϲ����Ľṹ��ͼ��ʾ�����й���10-�ǻ�ϲ�����˵����ȷ���ǣ�������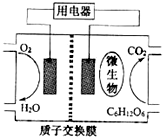 ��2009?���գ���������Ϊȼ�ϵ�����ȼ�ϵ�ؽṹʾ��ͼ��ͼ��ʾ�����ڸõ�ص�������ȷ���ǣ�������
��2009?���գ���������Ϊȼ�ϵ�����ȼ�ϵ�ؽṹʾ��ͼ��ͼ��ʾ�����ڸõ�ص�������ȷ���ǣ�������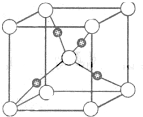
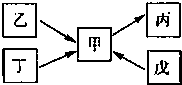 �ס��ҡ������������Ϊ��ѧ��ѧ�����ĵ��ʻ���Ҿ�����ͬһ��Ԫ��R����һ����������ת����ϵ��ͼ��ʾ��
�ס��ҡ������������Ϊ��ѧ��ѧ�����ĵ��ʻ���Ҿ�����ͬһ��Ԫ��R����һ����������ת����ϵ��ͼ��ʾ��