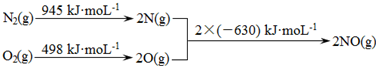��Ŀ����
2����֪1-������2-�����Ľṹ��ʽ���£�1-������
 ��2-������
��2-������ ��
��
��1����ͼ1�����������ʵ�����һ�ֵĺ˴Ź������ף����ҷ�����ȷֱ�Ϊ1��1��6���ݴ˿�֪��������2-�����������ƣ�������1-������2-������˵����ȷ����ABD������ѡ��
A��������ȥ��Ӧ������ͬ���� B����������ˮ��Ҳ�������л��ܼ�
C��2-������1-������Ϊͬϵ�� D�������Է������Ӽ���ˮ����
��֪��1-�����е㣺71��1-�����е㣺97.1�������ѷе㣺90��
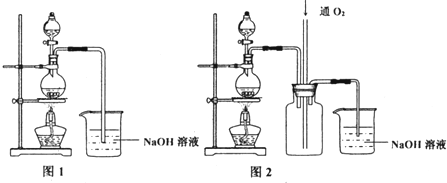
ʵ���ҿ���ͼ2װ���Ʊ�1-����飮��1-������Ũ���ᡢ�廯��һ����ȣ����뼸����ʯ��������69-72���������2h�������ռ�68-90�����Һ����̼������Һϴ�����ԣ��������ռ�68-76�����Һ��1-����飮
��2��Aװ������������ƿ��
��3���������л���Ӧ�Ļ�ѧ����ʽΪCH3CH2CH2OH+HBr$��_{��}^{ŨH_{2}SO_{4}}$CH3CH2CH2Br+H2O��
��4��Ϊ�˷�ֹ��������࣬��ʵ�����������Ũ������ǣ�C��
A.10% B.30% C.70% D.98.3%
��5��Ҳ�������ñ�ϩ��HBr�ӳ����Ʊ�����ϩ��ʯ���ѽ�IJ����ֱ����ԭ�ϣ�
���� ��1�����ݸ��л���ĺ˴Ź������еķ�����ȿ���ȷ�����л�������к��еIJ�ͬ������ԭ�ӵ�����������ȷ���л���Ľṹ��ʽ��1-������2-����������ȥ��Ӧ�����ɱ�ϩ����������ˮ��Ҳ�������л��ܼ�����Ϊͬ���칹�壬�����Է������Ӽ���ˮ���ѣ�
��2������װ��ͼ��֪Aװ�����ƣ�
��3��1-�������廯�ƿ���Ũ����������¼��ȿ�����1-����飻
��4��1-�������廯�Ʒ�ӦҪ��Ũ��������������98.3%��Ũ������ǿ���������ɣ������Ҵ���Ӧ���ɶ�����̼�Ͷ������ݴ��жϣ�
��5��ʯ���ѽ�ɵ���ϩ�ͱ�ϩ��С���Ӳ���������
��� �⣺��1�����ݸ��л���ĺ˴Ź������еķ�����ȷֱ�Ϊ1��1��6��֪�����л�������к��еIJ�ͬλ����ԭ����3�֣��ֱ�����ԭ����֮��Ϊ1��1��6��1-������CH3-CH2-CH2-OH}�в�ͬλ�õ���ԭ�������֣�����Ŀ֮��Ϊ1��3��4��2-���� �в�ͬλ�õ���ԭ�������֣�����ԭ����Ŀ֮��Ϊ1��1��6��������������������Ϊ��2-������1-������2-����������ȥ��Ӧ�����ɱ�ϩ����������ˮ��Ҳ�������л��ܼ�����Ϊͬ���칹�壬�����Է������Ӽ���ˮ���ѣ���ѡABD��
�в�ͬλ�õ���ԭ�������֣�����ԭ����Ŀ֮��Ϊ1��1��6��������������������Ϊ��2-������1-������2-����������ȥ��Ӧ�����ɱ�ϩ����������ˮ��Ҳ�������л��ܼ�����Ϊͬ���칹�壬�����Է������Ӽ���ˮ���ѣ���ѡABD��
�ʴ�Ϊ��2-������ABD��
��2������װ��ͼ��֪Aװ������Ϊ������ƿ��
�ʴ�Ϊ��������ƿ��
��3��1-�������廯�ƿ���Ũ����������¼��ȿ�����1-����飬��Ӧ�Ļ�ѧ����ʽΪCH3CH2CH2OH+HBr$��_{��}^{ŨH_{2}SO_{4}}$CH3CH2CH2Br+H2O��
�ʴ�Ϊ��CH3CH2CH2OH+HBr$��_{��}^{ŨH_{2}SO_{4}}$CH3CH2CH2Br+H2O��
��4��1-�������廯�Ʒ�ӦҪ��Ũ��������������98.3%��Ũ������ǿ���������ɣ������Ҵ���Ӧ���ɶ�����̼�Ͷ����������Ա�ʵ�����������Ũ�������70%��
��ѡC��
��5��ʯ���ѽ�ɵ���ϩ�ͱ�ϩ��С���Ӳ������������Ա�ϩ��ʯ���ѽ�IJ��
�ʴ�Ϊ���ѽ⣮
���� ���⿼�����л������ʽ��ȷ��֪ʶ����Ŀ�ѶȽ�С����ȷ�˴Ź������еķ�����ȿ���ȷ�����л�������к��еIJ�ͬ������ԭ�ӵ������ǽ���ؼ�������ͨ���������ѵ���ȡ�������˶���Ϣ�Ĵ����������Ի���ʵ�����������������Ѷ��еȣ�

| A�� | �ù��˵ķ�������CaCO3��CaCl2��Һ�Ļ���� | |
| B�� | �ýᾧ���ᴿNaCl��KNO3������е�KNO3 | |
| C�� | ���������Ҵ���ˮ�Ļ���� | |
| D�� | �ü��ȷ��������Ȼ�淋Ļ�����ʾ�������������Ȼ�������ֽ⣮��ȴ�����Ƕ����������ɹ��壩 |
| A�� | 1molH2����Ϊ2g | B�� | H2O��Ħ������Ϊ18g | ||
| C�� | �����44g CO2�����Ϊ22.4L | D�� | 9.8g H2SO4��1mol��H2SO4���� |
| A�� | 5min��ƽ����Ӧ����vA=0.15mol•L-1•min-1 | |
| B�� | ��ͬʱ��ԭƽ����ϵ����һ���¶Ⱥ�����һ��ѹǿʱ����ƽ���C�����ʵ�������Ϊ60%����÷�Ӧ������ӦΪ���ȷ�Ӧ | |
| C�� | ���������������䣬����ʼʱ�������м���0.5molA��0.5molB��1.5molC����Ӧ�����淴Ӧ������� | |
| D�� | t�棬��2L�ܱա����������м�������ʵ�����A��B����Ӧ�ﵽƽ��״̬ʱ��C�����ʵ���������Ϊ60%�������A�����ʵ���Ϊ1.6mol |
 25��ʱ������ƽ�ⳣ����
25��ʱ������ƽ�ⳣ����| ��ѧ�� | CH3COOH | H2CO3 | HClO |
| ����ƽ�ⳣ�� | 1.8��10-5 | K14.3��10-7 K25.6��10-11 | 3.0��10-8 |
��l�����ʵ���Ũ�Ⱦ�Ϊ0.1mol•L-l������������Һ��
a��Na2CO3��Һ��b��NaClO��Һ��c��CH3COONa��Һ��d��NaHCO3��Һ��
pH�ɴ�С��˳����A��B��D��C�����ţ���
��2��������0.1mol•L-l��CH3COOH��Һ��ˮϡ���̣����б���ʽ������һ����С����A
A��c��H+�� B��$\frac{c��{H}^{+}��}{c��C{H}_{3}COOH��}$ C��c��H+��•c��OH-�� D��$\frac{c��O{H}^{-}��}{c��{H}^{+}��}$
��3�����Ϊ10mLpH=2�Ĵ�����Һ��һԪ��HX�ֱ��ˮϡ����1000mL��ϡ������pH�仯����ͼ��HX�ĵ���ƽ�ⳣ�����ڣ�����ڡ��������ڡ���С�ڡ��������ƽ�ⳣ����������pH��ͬ�Ĵ����HXϡ����ͬ�ı�����HX��pH�仯��ϡ�ͺ�HX��Һ��ˮ���������c��H+������ ������Һ��ˮ���������c��H+����������ڡ��������ڡ���С�ڡ���������ϡ�ͺ������������Ũ�ȴ���HX�����Դ�������ˮ����̶ȴ���HX��
��4��25��ʱ��CH3COOH��CH3COONa�Ļ����Һ������û��ҺpH=6������Һ��c��CH3COO-��-c��Na+��=9.9��10-7mol/L����ȷ��ֵ����
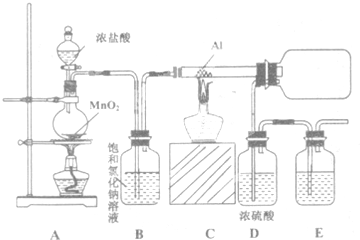
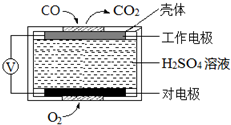 ̼��������������������Ĵ�������������������о����ȵ����⣮
̼��������������������Ĵ�������������������о����ȵ����⣮