��Ŀ����
��12�֣�A��B��C��D��E��F�����ֶ�����Ԫ�أ����ǵ�ԭ�������������� A��D��C��FΪͬ����Ԫ�أ�A�ĵ�������������壻 BԪ������������ˮ����������̬�⻯�ﷴӦ�����Σ� EԪ���ǵؿ��к������Ľ���Ԫ�ء�FԪ��ԭ��M���ϵ�������L����2�����ӡ�������������
��1��EԪ��λ�����ڱ��ĵ� ���� �塣
��2��F�������ӵĽṹʾ��ͼ ��
��3��A��C��D ����Ԫ�ع�ͬ�γɵĻ�����ĵ���ʽ ����������ڵĻ�ѧ���� ��
��4��ͭ��BԪ������������ˮ�����ϡ��Һ��Ӧ�����ӷ���ʽΪ ��
��5��E����������Һ�е���BԪ����̬�⻯���ˮ��Һ��ֱ�����������ӷ���ʽΪ ��
��1��3����A��2�֣���2��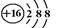 ��2�֣�
��2�֣�
��3��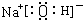 �����Ӽ����ۼ���2��+2�֣�
�����Ӽ����ۼ���2��+2�֣�
��4��3Cu��8H��+2NO3��=3Cu2����2NO����4H2O��2�֣�
��5��Al3+��3NH3��H2O= 3NH4+��Al(OH)3����2�֣�
����

��ϰ��ϵ�д�
 ���Ž�������С״Ԫϵ�д�
���Ž�������С״Ԫϵ�д�
�����Ŀ
��A��B��C��D��E��F��ƿ��ɫ��Һ�����Ƕ�����ѧ��ѧ�г��������Լ���E��������һ����ɫ��״Һ�壬B��C��D��F������Һ�������ǵ������Ӿ���ͬ���ֽ�������ʵ�飺
��A�д̼�����ζ����մ��Ũ����IJ������ӽ�Aʱ������ɫ����
�ڽ�AΪ�ֱ��������������Һ�У�ֻ��D��F���г��������������������Aʱ��D�г����ޱ仯��F�г�����ȫ�ܽ⣻
�۽�B�ֱ����C��D��E��F�У�C��D��F�в���������E������ɫ����ζ�����ݳ���
�ܽ�C�ֱ����D��E��F�У����г������ɣ��ټ���ϡHNO3���������ܣ�
��������ʵ����Ϣ������˵��������ǣ�������
��A�д̼�����ζ����մ��Ũ����IJ������ӽ�Aʱ������ɫ����
�ڽ�AΪ�ֱ��������������Һ�У�ֻ��D��F���г��������������������Aʱ��D�г����ޱ仯��F�г�����ȫ�ܽ⣻
�۽�B�ֱ����C��D��E��F�У�C��D��F�в���������E������ɫ����ζ�����ݳ���
�ܽ�C�ֱ����D��E��F�У����г������ɣ��ټ���ϡHNO3���������ܣ�
��������ʵ����Ϣ������˵��������ǣ�������
| A��B������NaHCO3��Һ | B��FΪAgNO3��Һ | C��D��������þ������������ | D��AΪ��ˮ��EΪ���� |
