��Ŀ����
��10�֣�2010���Ϻ������������ǡ����У�����������á����Իش��������⣺
��1��Ϊȷ���������ڼ���ǿ������������ʴﵽ95%���ϣ��������ڼ�Ŀ�������״������У�����Ҫ����ָ����_____________��
a������������PM10�� b��NO2Ũ�� c��SO2Ũ�� d��CO2Ũ�ȣ�
��2����ɳ�����ն�����Ҫ������_______________��Ϊ�˸��ƿ���������������ƴ����ж�������������̳�����Ⱦ����ŷ�������������װβ��������װ�ã�ʹ���е��к�����NO��COת��Ϊ�����壬�÷�Ӧ�Ļ�ѧ����ʽΪ__________��
��3��Ϊ2010���������ڼ��ṩ��ѧ��Ч�Ļ����������Ϸ�������������ˮ�������������죬������ˮʱ����K2SO4��Al2(SO4)3��24H2O���ʽ�Ȼ�����������____________��ͨ�������������ȵ�������______________��ijũ�����Ϊ�������ˮ���ڽ��ر�ˮȡ�ؼҺ�ʹ��Ư�ۻ�Ư��Ƭ����ɱ����������ԭ�����û�ѧ����ʽ��ʾΪ_________________________��
��4���������й��ݡ���������֮�ڡ������ɸֽ��������7000����ɫ�����1200��鲣���Ƚ��ɡ������������õĹ�ҵ�豸�� ��ʯӢ������ѧ�ȶ���ǿ������ϵ��С����һ�����ֲ�����ʯӢ��������Ҫ�ɷ��� ��
��10�֣���1��d
(2)���ﰺ ���ȷ���������������ȣ�2NO+2CO N2+2CO2
N2+2CO2
(3)����������������Ca(ClO)2+H2O+CO2=CaCO3��+2HClO
��4������Ҥ��������¯����SiO2
��������

 2010���Ϻ������᳡�ݣ��������������ϻ���Ļ��ʹ���˷�������ܣ�LED����Ŀǰ����LEDƷƬ�����ʻ�����GaAs���黯�أ���AlGaInP��������������lnGaN���������أ�Ϊ������֪������ͬ����һ���ڵ�Ԫ�أ��黯�صľ����ṹ��ͼ���Իش�
2010���Ϻ������᳡�ݣ��������������ϻ���Ļ��ʹ���˷�������ܣ�LED����Ŀǰ����LEDƷƬ�����ʻ�����GaAs���黯�أ���AlGaInP��������������lnGaN���������أ�Ϊ������֪������ͬ����һ���ڵ�Ԫ�أ��黯�صľ����ṹ��ͼ���Իش� 2010���Ϻ������᳡�ݴ������������ϻ���Ļ��ʹ���˷�������ܣ�LED����Ŀǰ����LED��Ƭ���ʻ�����GaAs���黯�أ���AlGaInP��������������InGaN���������أ�Ϊ������֪������ͬ����һ���ڵ�Ԫ�أ��黯�صľ����ṹ��ͼ���Իش�
2010���Ϻ������᳡�ݴ������������ϻ���Ļ��ʹ���˷�������ܣ�LED����Ŀǰ����LED��Ƭ���ʻ�����GaAs���黯�أ���AlGaInP��������������InGaN���������أ�Ϊ������֪������ͬ����һ���ڵ�Ԫ�أ��黯�صľ����ṹ��ͼ���Իش�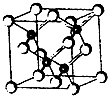 ��2011?�Ͼ�ģ�⣩2010���Ϻ������᳡�ݣ��������������ϻ���Ļ��ʹ���˷�������ܣ�LED����Ŀǰ����LEDƷƬ�����ʻ�����GaAs���黯�أ���AlGaInP��������������InGaN���������أ�Ϊ�����黯�ص�Ʒ���ṹ��ͼ���Իش�
��2011?�Ͼ�ģ�⣩2010���Ϻ������᳡�ݣ��������������ϻ���Ļ��ʹ���˷�������ܣ�LED����Ŀǰ����LEDƷƬ�����ʻ�����GaAs���黯�أ���AlGaInP��������������InGaN���������أ�Ϊ�����黯�ص�Ʒ���ṹ��ͼ���Իش�