��Ŀ����
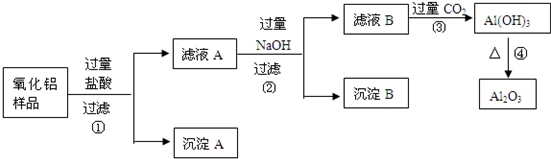
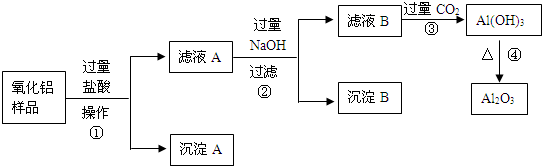
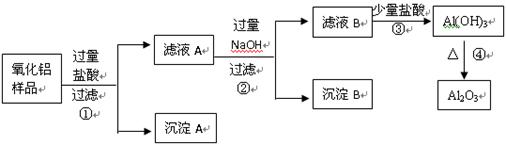
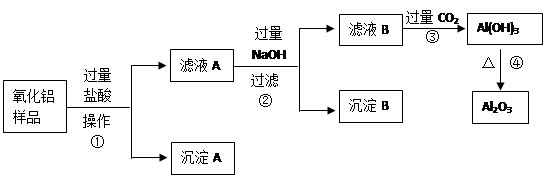
��6�֣�ij��������Ʒ�к����������Ͷ����������ʣ�������ȡ��������������ijͬѧ������µ�ʵ�鷽�����ش��������⣺
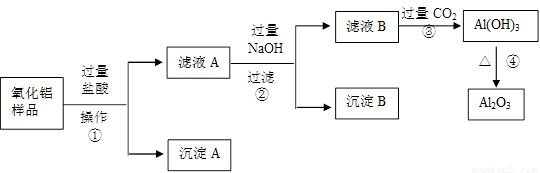
��1�������ٵ����� ��
��2������A�Ļ�ѧʽ��___________����ҺB�е������ӳ���Cl����OH�����______________��
��3������ܵĻ�ѧ����ʽΪ______________________________________��
��4���ڲ�������Ҫ�õ��IJ������������ձ���������������_________________��
���������������費�������ᣬͨ�����˼��õ�����A����Һ�к��е��ǹ��������ᣬ���ɵ��Ȼ������Ȼ������������������������Һ���Ȼ����������������������Ȼ�������ƫ�����ƣ����˼��õ�����������������ʱ��Һ�к��й��˵��������ƺ�����ƫ�������Լ��Ȼ��ƣ�ͨ������Ķ�����̼�����������������������������������ȶ������ȷֽ�������������
���𰸡�
��1������ ��2��SiO2 AlO2�� ��3��2Al(OH)3 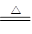 Al2O3
+ 3H2O ��4����ͨ©��
Al2O3
+ 3H2O ��4����ͨ©��

��ϰ��ϵ�д�
 ������ʱ����ҵ����ϵ�д�
������ʱ����ҵ����ϵ�д� ��ĩ���ƾ�ϵ�д�
��ĩ���ƾ�ϵ�д� ���ɿ��ñ���ϵ�д�
���ɿ��ñ���ϵ�д�
�����Ŀ