��Ŀ����
3��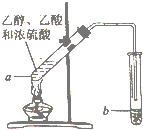 ����ͼ��ʾ��ʵ�װ���Ʊ�����������������ش��й����⣺
����ͼ��ʾ��ʵ�װ���Ʊ�����������������ش��й����⣺��1���Ҵ��з��ӹ����ŵ��������ǻ���
��2���Թ�a�м��뼸�����Ƭ��Ŀ���Ƿ�ֹҺ�屩�У�
��3���Թ�a�з�����Ӧ�Ļ�ѧ����ʽΪCH3COOH+CH3CH2OH
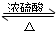 CH3COOC2H5+H2O����Ӧ������������Ӧ����ȡ����Ӧ����
CH3COOC2H5+H2O����Ӧ������������Ӧ����ȡ����Ӧ������4����Ӧ��ʼǰ���Թ�b��ʢ�ŵ���Һ�DZ���̼������Һ��
��5����Ӧ���������Թ�b�����ã��۲쵽��������b��Һ��ֲ㣬�ϲ���������״Һ�壮
��6�����÷�Һ�ķ������Ƶõ������������������
��7���Թ��еĵ��ܿ�ΪʲôҪ��Һ��֮�ϣ���ֹ������
���� ��1�����Ĺ�����Ϊ�ǻ���
��2���������Ƭ���ʯ���ɷ�ֹҺ��ɽ���
��3��������Ҵ���Ũ���������·���������Ӧ����������������ˮ��
��4���Ʊ���������ʱ���ñ���̼������Һ��������������Ŀ���dz�ȥ�Ҵ������ᡢ���������������ܽ�ȣ����ڷֲ㣻
��5��������������������̼������Һ�����ܶȽ�С���з�����
��6�����뻥�����ܵ�Һ��ͨ���÷�Һ�ķ�����
��7���ӷ�������������Ҵ�������ˮ����������������
��� �⣺��1���Ҵ��Ľṹ��ʽΪCH3CH2OH���京�еĹ�����Ϊ�ǻ���
�ʴ�Ϊ���ǻ���
��2�����Һ�е�ϵͣ����ȹ��������������У�����Ϊ��ֹҺ��ɽ���Ӧ�������Ƭ���ʯ��
�ʴ�Ϊ����ֹҺ�屩�У�
��3���������Ҵ���Ũ���������¼��ȷ���������Ӧ��������������ˮ��ͬʱ�÷�Ӧ�ǿ���ģ���Ӧ�Ļ�ѧ����ʽΪ��CH3COOH+CH3CH2OH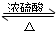 CH3COOC2H5+H2O���÷�ӦΪ������Ӧ��
CH3COOC2H5+H2O���÷�ӦΪ������Ӧ��
�ʴ�Ϊ��CH3COOH+CH3CH2OH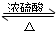 CH3COOC2H5+H2O��������Ӧ����ȡ����Ӧ����
CH3COOC2H5+H2O��������Ӧ����ȡ����Ӧ����
��4���Ʊ���������ʱ���ñ���̼������Һ����������������ȥ�Ҵ������ᡢ���������������ܽ�ȣ����ڷֲ㣬����b��ʢ�ŵ�Һ��Ϊ����̼������Һ��
�ʴ�Ϊ������̼������Һ��
��5���������������ڱ���̼������Һ�����ܶ�С��̼������Һ����b��Һ��ֲ㣬�ϲ���������״Һ�壬
�ʴ�Ϊ��b��Һ��ֲ㣬�ϲ���������״Һ�壻
��6����B�е�Һ������ֲ㲻���ܣ����÷�Һ�������룬
�ʴ�Ϊ����Һ��
��7��������Ҵ�������ˮ����������ϩ��������Һ������Ϊ�˷�ֹ������
�ʴ�Ϊ����ֹ������
���� ���⿼�������������Ʊ�����Ŀ�Ѷ��еȣ�ע��ʵ��Ļ������������Լ�Ũ������ʱ��ע�������ȷ������Ӧԭ��Ϊ���ؼ�������������ѧ���ķ�������ѧʵ��������

| A�� | ������O2��SO2��SO3�� | B�� | ֻ������O2��SO3�� | ||
| C�� | ֻ������O2��SO2�� | D�� | ֻ������O2�� |
| A�� | ��ǿ�Ͼ����ϵĻ��������ã�����Ч��������ɫ��Ⱦ | |
| B�� | �ϳ����ϡ��ϳ���ά���ϳ���������ϳɲ��� | |
| C�� | ���ͽṹ�����Ͼ��������� | |
| D�� | ���ϡ���ά�ء������ʡ���Ȼ��������Ȼ�߷��� |
| Ԫ�ر�� | Ԫ�����ʻ�ԭ�ӽṹ |
| T | �������������۵Ĵ�����Ϊ4 |
| X | �����������Ǵε�����������������2�� |
| Y | �����µ���Ϊ˫ԭ�ӷ��ӣ����⻯���ˮ��Һ�ʼ��� |
| Z | ���������+7�� |
��1��T��Ԫ�����ڱ��е�λ��Ϊ�������ڵڢ�A�壮
��2��X��һ��ͬλ�ؿ����ڲⶨ�����������ͬλ��Ϊ614C����ԭ�ӷ��ű�ʾ����
��3��Z���ʵĵ���ʽΪ
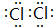 ��Y���⻯���ˮ��Һ�Լ��Ե�ԭ��ΪNH3+H2O?NH3•H2O?NH4++OH-�������ӷ���ʽ��ʾ����
��Y���⻯���ˮ��Һ�Լ��Ե�ԭ��ΪNH3+H2O?NH3•H2O?NH4++OH-�������ӷ���ʽ��ʾ������4��Z��T��ȣ��ǽ����Խ�ǿ����Cl����Ԫ�ط��ű�ʾ�������б�������֤����һ��ʵ����d����ѡ����ĸ��
a�������£�Z����Ϊ���壬T����Ϊ����
b��Z���⻯���ˮ��Һ�����Ա�Tǿ
c��Z��������۱�Y��
d������������Ӧˮ���������Z��Tǿ
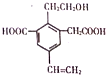
��5��Z���⻯����Y���⻯������Ϸ�Ӧ�������к��еĻ�ѧ������Ϊ���Ӽ������ۼ���
| A�� | 1mol�����ʣ������Լӳ�4molH2 | |
| B�� | ����������Na��NaOH��NaHCO3�����ʵ�����Ϊ3��2��2 | |
| C�� | ���������Ը��������Һ�������е�̼̼˫�� | |
| D�� | �������ܹ��ڴ��������±�����Ϊ��ȩ���Ļ����� |
| A�� | H+H��H-H | |
| B�� | Mg+2HCl�TMgCl2+H2�� | |
| C�� | 2NH4Cl+Ba��OH��2•8H2O�TBaCl2+2NH3��+10H2O | |
| D�� | H2SO4+2KOH�TK2SO4+2H2O |
 ��
��
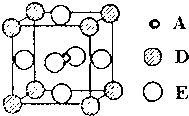 A��B��C��D��E����Ԫ�����ڱ���ǰ36�ŵ�Ԫ�أ����ǵ�ԭ������������Aԭ�Ӻ��������6�ֲ�ͬ���˶�״̬��C�Ļ�̬ԭ��2p�ܼ�����2��δ�ɶԵ��ӣ�C2-������D2+���Ӿ�����ͬ�ĵ��Ӳ�ṹ��E�Ļ�̬ԭ�ӵ���Χ�����Ų�ʽΪ3d84s2��
A��B��C��D��E����Ԫ�����ڱ���ǰ36�ŵ�Ԫ�أ����ǵ�ԭ������������Aԭ�Ӻ��������6�ֲ�ͬ���˶�״̬��C�Ļ�̬ԭ��2p�ܼ�����2��δ�ɶԵ��ӣ�C2-������D2+���Ӿ�����ͬ�ĵ��Ӳ�ṹ��E�Ļ�̬ԭ�ӵ���Χ�����Ų�ʽΪ3d84s2��