��Ŀ����
��12�֣�����AlCl3��FeCl3������к�������FeCl2��Al2��SO4��3��ijͬѧ��Ʋ�����ʵ��Ըû������з��룬�Եõ�������AlCl3��FeCl3��ͼ���Ǹ�ͬѧ��Ʋ����е�ʵ�飺
��1��ʵ���������������˵��Լ�������Ϊ���ѡ�õ��Լ���___________��
A��KMnO4��H������Һ B������ C��H2O2 D��Ũ����
��2������V�����ӷ���ʽΪ_______________________________________________��
�����������ӷ���ʽΪ________________________________________________��
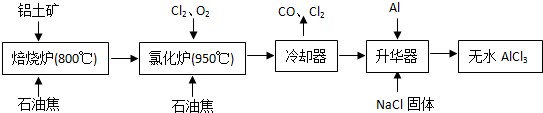
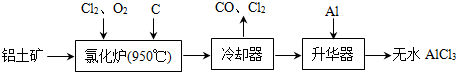
��3��ͼ���Dz�������X��ȡ��ˮFeCl3��AlCl3�IJ���װ�ã�����̨������װ�õ�����ȥ����
��ͼ���У�װ��a��____________��Բ����ƿ��˫�����͵�����ɡ�
��ѭ������A��______________________��
��װ��b���������ʿ�����_______________________�����ţ���
A����ʯ�� B����ˮ�Ȼ��� C��Ũ���� D������������
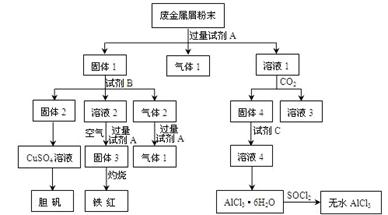
��4������Ϊ��ͬѧ��ʵ�����_____________����ܡ����ܡ����õ�������AlCl3��FeCl3�������ܣ��������Ƹ�ͬѧ��ʵ�鲽����ƣ���������������________________
_____________________________________________�������ܣ��˿ղ��
����:

 ��Ȥ������ҵ���ϿƼ�������ϵ�д�
��Ȥ������ҵ���ϿƼ�������ϵ�д�