��Ŀ����
ij��ѧ�о���ѧϰС���ij��ɫˮ���ijɷֽ��м��飬��֪��ˮ����ֻ���ܺ���K+��Mg2+��Fe3+��Cu2+��Al3+��Ag+��Ca2+��CO
��SO
��Cl-�е����������ӣ���С��ͬѧȡ100mLˮ������ʵ�飬��ˮ�����ȵμ����ᱵ��Һ���ٵμ�1mol/L�����ᣬʵ������г��������ı仯�������ͼ��ʾ��
��1��ˮ����һ�����е���������______�������ʵ���Ũ��֮��Ϊ______��
��2��д��BC������ʾ��Ӧ�����ӷ���ʽ��______��
��3����B��C��仯������������������Ϊ______��
��4���Ը���ʵ�����Ʋ�K+�Ƿ���ڣ�______����ǡ����������ڣ�K+�����ʵ���Ũ��c��K+���ķ�Χ��______������K+�����ڣ��ػش���ʣ���
��5����Ƽ�ʵ����֤ԭˮ���п��ܴ��ڵ����ӣ�______����д��ʵ�鲽�衢����ͽ��ۣ�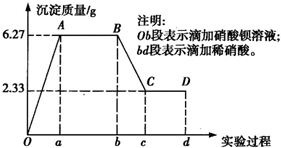
| 2-3 |
| 2-4 |
��1��ˮ����һ�����е���������______�������ʵ���Ũ��֮��Ϊ______��
��2��д��BC������ʾ��Ӧ�����ӷ���ʽ��______��
��3����B��C��仯������������������Ϊ______��
��4���Ը���ʵ�����Ʋ�K+�Ƿ���ڣ�______����ǡ����������ڣ�K+�����ʵ���Ũ��c��K+���ķ�Χ��______������K+�����ڣ��ػش���ʣ���
��5����Ƽ�ʵ����֤ԭˮ���п��ܴ��ڵ����ӣ�______����д��ʵ�鲽�衢����ͽ��ۣ�

��ˮ��Ϊ��ɫ��Һ��ˮ��Fe3+��Cu2+�����ڣ�����ͼ������������ᱵ��Һ���ɳ���������ϡ���ᣬ���������ܽ�֤��ˮ����һ������SO42-��CO32-������ΪAl3+��CO32-����˫ˮ�⣬CO32-��Ag+��Ca2+��Mg2+������Ӧ���ɳ������ܴ������ڣ�����Al3+��Ag+��Ca2+��Mg2+�����ڣ�n��SO42-��=
=0.01mol��n��CO32-��=n��BaCO3��=
=0.02mol��c��SO42-����c��CO32-��=1��2��ԭ��Һ�п��ܺ���Cl-�����ݵ������Һ�е���غ㣬������K+һ�����ڣ����ݵ���غ�õ���0.01mol��2+0.02mol��2+n��Cl-��=n��K+����֪n��K+����0.06mol����c��K+����0.6mol/L��
��1��ˮ����һ�����е���������SO42-��CO32-�������ʵ���Ũ��֮��Ϊ1��2��
�ʴ�Ϊ��SO42-��CO32-��1��2��
��2��BC������ʾ��Ӧ��̼�ᱵ����ϡ����ķ�Ӧ����Ӧ�����ӷ���ʽ��BaCO3+2H+=Ba2++CO2��+H2O���ʴ�Ϊ��BaCO3+2H+=Ba2++CO2��+H2O��
��3����B��C��仯����������ͼ�����������̼�ᱵn��BaCO3��=
=0.02mol������ϡ�������ʵ���Ϊ0.04mol��������������=
=
=0.04L=40ml���ʴ�Ϊ��40ml��
��4�����ݵ������Һ�е���غ㣬������K+һ�����ڣ����ݵ���غ�õ���0.01mol��2+0.02mol��2+n��Cl-��=n��K+����֪n��K+����0.06mol����c��K+����0.6mol/L���ʴ�Ϊ���ǣ���0.6mol/L��
��5�����ܴ��ڵ�������Cl-��ʵ�����Ϊ��ȡ����ˮ�����Թ��У����Թ��м�������������ᱵ��Һ��ϡ���ᣬ��������ȫ�����������ɺ����ϲ���Һ�еμ���������������Һ�������ɰ�ɫ��������ԭˮ���� ����Cl-�����ް�ɫ�������ɣ�֤����Cl-���ڣ�
�ʴ�Ϊ��ȡ����ˮ�����Թ��У����Թ��м�������������ᱵ��Һ��ϡ���ᣬ��������ȫ�����������ɺ����ϲ���Һ�еμ���������������Һ�������ɰ�ɫ��������ԭˮ���� ����Cl-�����ް�ɫ�������ɣ�֤����Cl-���ڣ�
| 2.33g |
| 233g/mol |
| 6.27g-2��33g |
| 197g/mol |
��1��ˮ����һ�����е���������SO42-��CO32-�������ʵ���Ũ��֮��Ϊ1��2��
�ʴ�Ϊ��SO42-��CO32-��1��2��
��2��BC������ʾ��Ӧ��̼�ᱵ����ϡ����ķ�Ӧ����Ӧ�����ӷ���ʽ��BaCO3+2H+=Ba2++CO2��+H2O���ʴ�Ϊ��BaCO3+2H+=Ba2++CO2��+H2O��
��3����B��C��仯����������ͼ�����������̼�ᱵn��BaCO3��=
| 6.27g-2��33g |
| 197g/mol |
| n |
| c |
| 0.04mol |
| 1mol/L |
��4�����ݵ������Һ�е���غ㣬������K+һ�����ڣ����ݵ���غ�õ���0.01mol��2+0.02mol��2+n��Cl-��=n��K+����֪n��K+����0.06mol����c��K+����0.6mol/L���ʴ�Ϊ���ǣ���0.6mol/L��
��5�����ܴ��ڵ�������Cl-��ʵ�����Ϊ��ȡ����ˮ�����Թ��У����Թ��м�������������ᱵ��Һ��ϡ���ᣬ��������ȫ�����������ɺ����ϲ���Һ�еμ���������������Һ�������ɰ�ɫ��������ԭˮ���� ����Cl-�����ް�ɫ�������ɣ�֤����Cl-���ڣ�
�ʴ�Ϊ��ȡ����ˮ�����Թ��У����Թ��м�������������ᱵ��Һ��ϡ���ᣬ��������ȫ�����������ɺ����ϲ���Һ�еμ���������������Һ�������ɰ�ɫ��������ԭˮ���� ����Cl-�����ް�ɫ�������ɣ�֤����Cl-���ڣ�

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
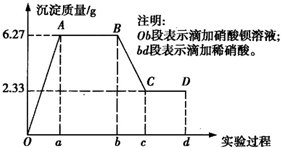 ij��ѧ�о���ѧϰС���ij��ɫˮ���ijɷֽ��м��飬��֪��ˮ����ֻ���ܺ���K+��Mg2+��Fe3+��Cu2+��Al3+��Ag+��Ca2+��CO
ij��ѧ�о���ѧϰС���ij��ɫˮ���ijɷֽ��м��飬��֪��ˮ����ֻ���ܺ���K+��Mg2+��Fe3+��Cu2+��Al3+��Ag+��Ca2+��CO
 ��2011?̩��һģ��ij��ѧ�о���ѧϰС��Ϊ̽��ijƷ�ƻ������в�����֬����ĺ���������������ʵ�飺
��2011?̩��һģ��ij��ѧ�о���ѧϰС��Ϊ̽��ijƷ�ƻ������в�����֬����ĺ���������������ʵ�飺
