��Ŀ����
��֪��A��B��C��D��E��F����Ԫ�غ˵�����������������ڱ���ǰ�����ڵ�Ԫ�ء�����Aԭ�Ӻ���������δ�ɶԵ��ӣ�������B2E�ľ���Ϊ���Ӿ��壬Eԭ�Ӻ����M����ֻ�����ԳɶԵ��ӣ�CԪ���ǵؿ��к�����ߵĽ���Ԫ�أ�D���ʵ��۵���ͬ����Ԫ���γɵĵ�������ߣ�F���γɺ�ɫ(��ש��ɫ)��F2O�ͺ�ɫ��FO���������
�ش��������⣺
��1��F��ԭ�ӵ�M������Ų�ʽΪ ��
��2��B��C��D�ĵ�һ��������С�����˳��Ϊ ��(��Ԫ�ط��ű�ʾ)
��3��A�ļ��⻯����Ӽ�������ˮ,����Ҫԭ���� .
��4��E�������������ӵĿռ乹���� ��������ԭ�ӵ��ӻ���ʽΪ ��
��5��F�ĸ�������A�ļ��⻯���γɵ�������,��λ��Ϊ ��
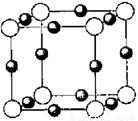
��6��A��F�γ�ij�ֻ�����ľ����ṹ��ͼ��ʾ�����仯ѧʽΪ ������ɫ���ʾFԭ�ӣ�,��֪���ڵİ��������֮��ľ���Ϊa cm, �þ������ܶ�Ϊ g/cm3��
��1��3s23p63d10��2�֣�
��2��Na��Al��Si��2�֣�
��3����������ˮ����֮����������2�֣�
��4��ƽ���������Σ�2�֣� sp2��1�֣�
��5�� 4��2�֣�
��6��Cu3N ��2�֣� 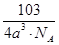 ��2�֣�������������Ҳ���֣�
��2�֣�������������Ҳ���֣�
��������
���������ԭ�Ӻ���������δ�ɶԵ��ӣ�������Ų�ʽΪ1S22S22P3��ΪNԪ�أ�Eԭ�Ӻ����M����ֻ�����ԳɶԵ��ӣ������Ų�ʽΪ1s22s22p3��ӦΪSԪ�أ�CԪ���ǵؿ��к�����ߵĽ���Ԫ�أ�ΪAlԪ�أ�������B2E�ľ���Ϊ���Ӿ��壬BӦΪ�ڢ�A��Ԫ�أ���ԭ��������NԪ�غ�Al֮�䣬ӦΪNaԪ�أ�D���ʵ��۵���ͬ����Ԫ���γɵĵ���������ߵģ�ӦΪSiԪ�أ����ʹ�Ϊԭ�Ӿ��壬�۵��ڵ�����������ߣ�F���γɺ�ɫ(��ש��ɫ)��F2O�ͺ�ɫ��FO���������ӦΪCuԪ�أ�
��1��Cu��ԭ�ӵĵ����Ų�ʽΪ1s22s22P63s23p63d104s1��M������Ų�ʽΪ3s23p63d10
��2����Ԫ�����ڱ��У�ͬһ����Ԫ�صĵ�һ�����ܴ�����������ͬһ����Ԫ�صĵ�һ�����ܴ��ϵ�����С���ݴ˿��ж�����Ԫ�صĵ�һ�����ܵ�˳��Ϊ��Na��Al��Si
��3��A��N���ļ��⻯������ǰ�����������������ˮ,����Ҫԭ��N��O�ĵ縺��ǿ������֮���γ������
��4��S03�к���3���ļ����µ��Ӷ���Ϊ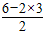 =0�����Է��ӵĿռ乹����ƽ���������Σ�sp2�ӻ�
=0�����Է��ӵĿռ乹����ƽ���������Σ�sp2�ӻ�
��5��ͭ�����백��������λ���γ������仯ѧʽΪ��[Cu(NH3)4]2+
��6�����ݾ������������ķ��䷽�����㣬�����к���ԭ�ӵ���ĿΪ8�� =1��ԭ�ӵ���ĿΪ��12��
=1��ԭ�ӵ���ĿΪ��12��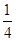 =3���ʻ�ѧʽΪCu3N�����������֮��ľ���Ϊa cm���߳�Ϊ2acm����
=3���ʻ�ѧʽΪCu3N�����������֮��ľ���Ϊa cm���߳�Ϊ2acm����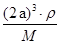 ��NA��1����æѣ�
��NA��1����æѣ�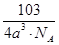 ��
��
���㣺����Ԫ�����ڱ��Ľṹ�Լ�Ԫ��ԭ�ӵĺ�������Ų�����λ��������ṹ�������۵��ж��Լ�������йؼ��㡣

 ���㼤�������100�ִ��Ծ�ϵ�д�
���㼤�������100�ִ��Ծ�ϵ�д� ��2011?�����ģ����ѧ--ѡ�����ʽṹ������
��2011?�����ģ����ѧ--ѡ�����ʽṹ������
 ��֪��A��B��C��D��E��F��XΪ���ڱ���ǰ�����ڵ�����Ԫ�أ����ǵ�ԭ��������������A�����ڱ���ԭ�Ӱ뾶��С��Ԫ�أ�B�Ļ�̬ԭ����3����ͬ���ܼ������ܼ��е�������ȣ�D�Ļ�̬ԭ��2p�ܼ��ϵ�δ�ɶԵ�������Bԭ�ӵ���ͬ��D2-������E2+���Ӿ�����ͬ���ȶ����Ӳ�ṹ��F�С����������֮�ƣ�F4+���Ӻ��ԭ�ӵĺ�������Ų���ͬ��X�Ļ�̬ԭ�ӵļ۵����Ų�ʽΪ3d84s2��
��֪��A��B��C��D��E��F��XΪ���ڱ���ǰ�����ڵ�����Ԫ�أ����ǵ�ԭ��������������A�����ڱ���ԭ�Ӱ뾶��С��Ԫ�أ�B�Ļ�̬ԭ����3����ͬ���ܼ������ܼ��е�������ȣ�D�Ļ�̬ԭ��2p�ܼ��ϵ�δ�ɶԵ�������Bԭ�ӵ���ͬ��D2-������E2+���Ӿ�����ͬ���ȶ����Ӳ�ṹ��F�С����������֮�ƣ�F4+���Ӻ��ԭ�ӵĺ�������Ų���ͬ��X�Ļ�̬ԭ�ӵļ۵����Ų�ʽΪ3d84s2��
 ��֪����A��B��C��D�����µķ�Ӧ��ϵ������A ��B��ȼ��ʱ������ʲ�ɫ�� C��B��ȼ��ʱ�����ػ�ɫ���̣�E��ˮ��Һ������ɫ��G��һ�ֺ�ɫ���壮
��֪����A��B��C��D�����µķ�Ӧ��ϵ������A ��B��ȼ��ʱ������ʲ�ɫ�� C��B��ȼ��ʱ�����ػ�ɫ���̣�E��ˮ��Һ������ɫ��G��һ�ֺ�ɫ���壮