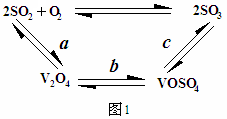
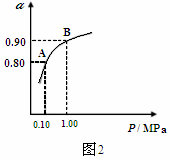
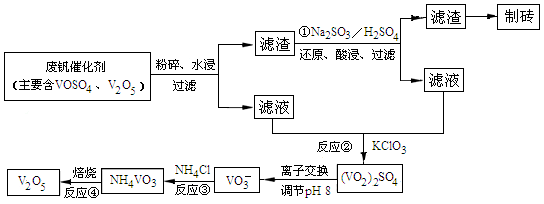
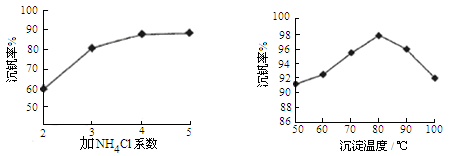
��Ŀ����
������ҵ�Դٽ����ú���ᷢչ������Ҫ���á�
(1)������¯��Ϊ�岿�֣�����ʯ�����ú����Ҫ��________���ַ�����Ӧ����________���ֿ�ʼ����������¯����
(2)����ʱ�����Ļ�ѧ����ʽΪ____________________������衢�̺�����Ŀ����________________________��
(3)����ֺ��е�CrԪ���������ֹ��̵�����________(�ǰ����)���룬ԭ����______________________��
(4)���������������У�β���о����е���Ҫ��Ⱦ����________���ӻ����;��ýǶȿ��ǣ�����β��������������________��
(1)¯����¯��
(2)2P��5FeO��3CaO Ca3(PO4)2��5Fe�������͵����ֵijɷ�
Ca3(PO4)2��5Fe�������͵����ֵijɷ�
(3)����Cr������(������ǰ����Cr���γ�¯������ȥ��)
(4)CO��ȼ��(��ԭ��)
����

����˵���У�����ȷ����
| A���������ܴӺ�ˮ����ȡ��ˮ |
| B���Ӻ�ˮ�п��Եõ��Ȼ�þ���ټ��ȷֽ���ƽ���þ |
| C���������Ӻ�ˮ������Ĺؼ���Ӧ��Cl2+2Br��= 2Cl��+Br2 |
D��ú��������Ҫ��Ӧ�� |
����ѧ��ѡ��2����ѧ�뼼������15�֣�
��̬��ҵ���Ľ��裬�����������ֻ��������Ҫ����ѭ���������ۺͳ�ֿ��Ǿ��õĿɳ�����չ��������ij��ҵ��Ƶ����ᣭ��泥�ˮ����������ˮ����ˮ���ã��Σ��ȣ�������������̬��ҵ������ͼ��
����������ҵ���̻ش��������⣺
��1����ԭ�ϡ���Դ����ͨ�Ƕȿ��Ǹ���ҵӦ���ڣ�������
����
| A������ɽ������ | B���غ��������� | C��������С��� | D��������½ |
��3������¯������Ӧ�Ļ�ѧ����ʽ�������������������������������� ,�ʳ��IJ�Ʒ���ոƣ�����Ҫ�ɷ��������������� (�ѧʽ)��
��4���ȵ糧����ȴˮ��������������������Ũ����ˮ����ȡ���������ȡ��������������д��һ�ּ��ɣ���
��5��������̬�����������������¯�������������������������ܵ��������������������������������������������������������� ����д�����㣩��
����ĽṹΪCH3��CH2��COOH,�������ǰ�ȫ��Ч�ķ�ù��������,һ���Լ�ʽ̼��пΪԭ�ϵ�����������������:
| ��� | n(����)�� n(��ʽ̼��п) | ��Ӧ�¶�/�� | ����п����/% |
| 1 | 1��0.25 | 60 | 67.2 |
| 2 | 1��0.25 | 80 | 83.5 |
| 3 | 1��0.25 | 100 | 81.4 |
| 4 | 1��0.31 | 60 | 89.2 |
| 5 | 1��0.31 | 80 | 90.1 |
| 6 | 1��0.31 | 100 | 88.8 |
(1)̽����ʵ������ѹ�������(���ϱ�):��Ӧʱ��2 h,��ˮ��45 g,n(����)��n(��ʽ̼��п)=1��
 ,��Ӧ�¶����������档
,��Ӧ�¶����������档 (2)�����ղ��á���·ѭ������ʽ,�������Ʊ����ռ�㡢���ʸ���,�������������������������������������������ŵ㡣
(3)ij��ʵ��ʱ,��37.0 g��������220 mLˮ��,�����������������Ż����������Ʊ�,���յñ���п49.6 g,��ô�ʵ�����п�IJ���Ϊ��������(д���������)��
����ĽṹΪCH3��CH2��COOH,�������ǰ�ȫ��Ч�ķ�ù��������,һ���Լ�ʽ̼��пΪԭ�ϵ�����������������:
| ��� | n(����)�� n(��ʽ̼��п) | ��Ӧ�¶�/�� | ����п����/% |
| 1 | 1��0.25 | 60 | 67.2 |
| 2 | 1��0.25 | 80 | 83.5 |
| 3 | 1��0.25 | 100 | 81.4 |
| 4 | 1��0.31 | 60 | 89.2 |
| 5 | 1��0.31 | 80 | 90.1 |
| 6 | 1��0.31 | 100 | 88.8 |
(1)̽����ʵ������ѹ�������(���ϱ�):��Ӧʱ��2 h,��ˮ��45 g,n(����)��n(��ʽ̼��п)=1��
 ,��Ӧ�¶����������档
,��Ӧ�¶����������档 (2)�����ղ��á���·ѭ������ʽ,�������Ʊ����ռ�㡢���ʸ���,���������������������������ŵ㡣
(3)ij��ʵ��ʱ,��37.0 g��������220 mLˮ��,�����������������Ż����������Ʊ�,���յñ���п49.6 g,��ô�ʵ�����п�IJ���Ϊ��������(д���������)��
���������LiFePO4��һ��������������ӵ�صĵ缫���ϡ�ij�����������졢﮻�ʯLiAl��SiO3��2��������Ca2+��Mg2+���Σ���̼�۵�ԭ����������������ﮡ�����Ҫ�����������£�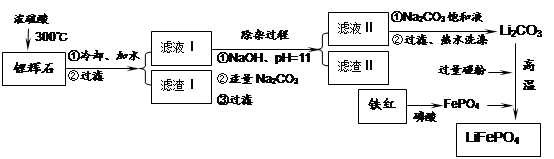
��֪��2LiAl��SiO3��2 + H2SO4(Ũ) 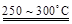 Li2SO4 + Al2O3��4SiO2��H2O��
Li2SO4 + Al2O3��4SiO2��H2O��
| �¶�/�� | 20 | 40 | 60 | 80 |
| �ܽ��(Li2CO3)/g | 1.33 | 1.17 | 1.01 | 0.85 |
| �ܽ��(Li2SO4)/g | 34.2 | 32.8 | 31.9 | 30.7 |
��1�����������пɷ����Al2O3������ͼ��ʾ����д�����ɳ��������ӷ���ʽ ��
��2�����������Ҫ�ɷ��ǣ� ���ѧʽ����
��3������Һ���м��뱥��Na2CO3��Һ�����˺��á���ˮϴ�ӡ���ԭ����
��
��4��д���ڸ�����������������﮵Ļ�ѧ����ʽ ��
��5����������﮵���ܷ�ӦΪ��FePO4+Li
 LiFePO4������еĹ������ʿɴ���Li������д���õ�طŵ�ʱ��������Ӧ�� �����øõ�ص�ⱥ��ʳ��ˮ�����ص缫��Ϊ���Ե缫������������������4480mL���壨��״��������ʱ���õ������﮵�����Ϊ ��
LiFePO4������еĹ������ʿɴ���Li������д���õ�طŵ�ʱ��������Ӧ�� �����øõ�ص�ⱥ��ʳ��ˮ�����ص缫��Ϊ���Ե缫������������������4480mL���壨��״��������ʱ���õ������﮵�����Ϊ �� ������(AlN)�������¡�������������Ժõ��������ʣ����㷺Ӧ���ڵ��ӹ�ҵ���մɹ�ҵ��������һ�������£���������ͨ�����·�Ӧ�ϳɣ�Al2O3��N2��3C=2AlN��3CO������������ȷ����(����)��
| A���ڵ������ĺϳɷ�Ӧ�У�N2�ǻ�ԭ����Al2O3�������� |
| B��������Ӧ��ÿ����2 mol AlN��N2�õ�3 mol���� |
| C���������е�Ԫ�صĻ��ϼ�Ϊ��3�� |
| D�����������ڸ��ϲ��� |