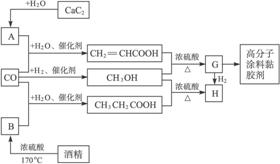
��Ŀ����
CO�����Ǽ���ú������Ҫ�ɷݣ�Ҳ����Ҫ�Ļ���ԭ�ϡ�����������������һ�ֵ��µ�ѹ�����գ���ijЩ���л��ᆳ���ʻ�����Ӧ�����������һ������������ܵ�װ���Ը߷���Ϳ�ϡ�ճ�����ȡ�����ͼ��ʾ��

ͼ��G��RCOOR/����һ��ͬ���칹����E������ͬϵ���H��һ��ͬ���칹������F������ͬϵ���֪D��CO��H2�����ʵ���֮��Ϊ1��2��ȫ��Ӧ���ɣ���������ɷ���������Ӧ��H�Ǻ���4��̼ԭ�ӵĻ������д����
��1���ṹ��ʽ��E_____________��G______________��R/��______________��
��2��G������ͬ���ͬ���칹��Ľṹ��ʽ������R��ĸ��_________��__________��
��3����Ӧ���ͣ�X_____________��Y_________________��Z_________________��
��4��������л�ѧ����ʽ��
A+CO+H2O![]() E��______________________________________________��
E��______________________________________________��
F+D![]() H��_________________��
H��_________________��
��1��E��CH2=CH�DCOOH��G��CH2=CH�DCOOCH3��R/�����DCH3��
��2��HCOOCH2�DCH=CH2��CH3COOCH=CH2��
��3��X���ӳɷ�Ӧ��Y����ȥ��Ӧ��Z���Ӿ۷�Ӧ��
��4��HC��CH+CO+H2O![]() H2C=CH�DCOOH
H2C=CH�DCOOH
H3C-CH2-COOH+CH3OH![]() H3C�DCH2�DCOOCH3+H2O��
H3C�DCH2�DCOOCH3+H2O��

 һ��һ����ʱ���ϵ�д�
һ��һ����ʱ���ϵ�д���14�֣�CO�����Ǽ���ú������Ҫ�ɷ֣�Ҳ����Ҫ�Ļ���ԭ�ϡ�����������������һ�ֵ��µ�ѹ�����գ���ijЩ���л��ᆳ���ʻ�����Ӧ�����������һ������������ܵ�װ���Ը߷���Ϳ�ϡ�ճ�ϼ��ȡ�����ͼ��ʾ��
 |
ͼ��G(RCOOR��)��һ��ͬ���칹����E������ͬϵ���H��һ��ͬ���칹������F������ͬϵ���֪D��CO��H2�����ʵ���֮��Ϊ1��2��ȫ��Ӧ���ɣ�����������ɷ���������Ӧ��H�Ǻ���4��̼ԭ�ӵĻ������д����
��1���ṹ��ʽ��E ��G ��R���� ��
��2��G������ͬ���ͬ���칹��Ľṹ��ʽ������R��ĸ�� �� ��
��3����Ӧ���ͣ�X ��Y ��Z ��

 ��4��д������ת���Ļ�ѧ����ʽ��
��4��д������ת���Ļ�ѧ����ʽ���� A + CO + H2O E�� ��F + D H
�� ��
�� ��