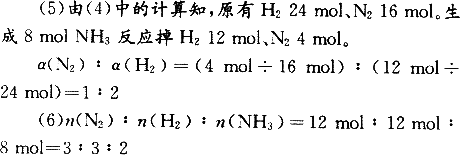��Ŀ����
�����£���a mol��N2��b mol��H2�Ļ������ͨ��һ���̶��ݻ����ܱ������У��������·�Ӧ��N2(g)��3H2(g)![]() 2NH3(g)��
2NH3(g)��
(1)����Ӧ��ijʱ��tʱ��nt(N2)��13 mol��nt(NH3)��6 mol������a��ֵ��
(2)��Ӧ��ƽ��ʱ�������������Ϊ716.8 L(��״����)������NH3�ĺ���(�������)Ϊ25��������ƽ��ʱNH3�����ʵ�����
(3)ԭ���������ƽ��������������ʵ���֮��(д����������ȣ���ͬ)n(ʼ)��n(ƽ)��________��
(4)ԭ��������У�a��b��________��
(5)�ﵽƽ��ʱ��N2��H2��ת����֮�Ȧ�(N2)�æ�(H2)��________��
(6)ƽ���������У�n(N2)��n(H2)��n(NH3)��________��
�𰸣�
������
��ʾ��
������
|
|
��ʾ��
|
(����ָ��)�����Ƚϸ��ӵ�ƽ������⣬Ҫ�Ƚ�������Ū����ֹ�ϵ�������н�����ѧƽ��ģʽ����һ�ֺܺõķ����� |

��ϰ��ϵ�д�
 ÿ�α���ϵ�д�
ÿ�α���ϵ�д�
�����Ŀ