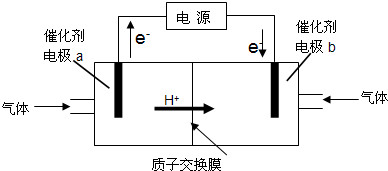
��Ŀ����
�״�-����ȼ�ϵ�أ�DMFC����һ�ָ�Ч�ܡ�����Ⱦ�綯�����ij��ص�أ���ȼ�ϵ�صĵ�ط�ӦʽΪ��CH3OH��l��+
O2��g���TCO2��g��+2H2O��l�����乤��ԭ��ʾ��ͼ���£�
��ش��������⣺
��1��д����ͼ��b��c�������ͨ����������ƣ���ѧʽ��b
��2�������ĵ缫��ӦʽΪ
��3���ø�ԭ��ص��AgNO3��Һ����Fe�缫����5.4g����ȼ�ϵ�������������ĵ����������Ϊ
| 3 | 2 |

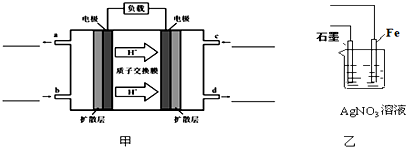
��ش��������⣺
��1��д����ͼ��b��c�������ͨ����������ƣ���ѧʽ��b
�״�
�״�
c����
����
��2�������ĵ缫��ӦʽΪ
CH3OH+H2O-6e-�TCO2+6H+
CH3OH+H2O-6e-�TCO2+6H+
����3���ø�ԭ��ص��AgNO3��Һ����Fe�缫����5.4g����ȼ�ϵ�������������ĵ����������Ϊ
280
280
mL����״������������1�����������ӵ��ƶ�����֪���ұߵ缫����������ߵ缫�Ǹ�����������ȼ��ʧ���ӷ���������Ӧ���������������õ��ӷ�����ԭ��Ӧ���ݴ��ж�b��c�缫�ϵ����ʣ�
��2�������ϼ״�ʧ���Ӻ�ˮ��Ӧ���ɶ�����̼�������ӣ�
��3������ת�Ƶ����غ���㣮
��2�������ϼ״�ʧ���Ӻ�ˮ��Ӧ���ɶ�����̼�������ӣ�
��3������ת�Ƶ����غ���㣮
����⣺��1�����������ӵ��ƶ�����֪���ұߵ缫����������ߵ缫�Ǹ�����������Ͷ��ȼ�ϼ״���������Ͷ���������������ʴ�Ϊ���״���������
��2�������ϼ״�ʧ���Ӻ�ˮ��Ӧ���ɶ�����̼�������ӣ��缫��ӦʽΪ��CH3OH+H2O-6e-�TCO2+6H+���ʴ�Ϊ��CH3OH+H2O-6e-�TCO2+6H+��
�۵����������Һ�У�����������������������������������ת�Ƶ����غ�֪�����������=
=0.28L=280mL���ʴ�Ϊ��280��
��2�������ϼ״�ʧ���Ӻ�ˮ��Ӧ���ɶ�����̼�������ӣ��缫��ӦʽΪ��CH3OH+H2O-6e-�TCO2+6H+���ʴ�Ϊ��CH3OH+H2O-6e-�TCO2+6H+��
�۵����������Һ�У�����������������������������������ת�Ƶ����غ�֪�����������=
| ||
| 4 |
���������⿼��ȼ�ϵ�أ���ȷȼ�ϵ�����������ϵ�ʧ�����ǽⱾ��ؼ����ѵ�����д�缫��Ӧʽ��ע��Ҫ��ϵ������Һ���������д��Ϊ�״��㣮

��ϰ��ϵ�д�
 ѧ�ڸ�ϰһ��ͨѧϰ�ܶ�Ա��ĩ������ӱ����������ϵ�д�
ѧ�ڸ�ϰһ��ͨѧϰ�ܶ�Ա��ĩ������ӱ����������ϵ�д�
�����Ŀ
�����������������ƽ�ȼ�ϵ�ص��з���ȼ�ϵ�ص�ȼ�����������״��ȣ�
��1��ͨ��������--��ѭ�����տ��Ƶ�ȼ�ϵ�ص�ȼ��������
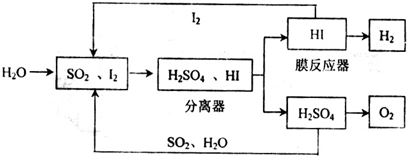
�ٸ�ѭ�����չ��̵��ܷ�Ӧ����ʽΪ ��
���û�ѧƽ���ƶ���ԭ����������HI�ֽⷴӦ��ʹ��Ĥ��Ӧ�������H2��Ŀ����
��2��������̼�ǵ�������ЧӦ��������ף�Ŀǰ���Ǵ���������̼�ķ���֮һ��ʹ����������Ӧ�ϳɼ״�����֪�������״�ȼ�յ��Ȼ�ѧ����ʽ���£�
2H2��g��+O2��g��=2H2O��l������H=-283.0kJ?mol-1����
2CH3OH��1��+3O2��g����2CO2��g��+4H2O��1������H=-726.0kJ?mol-1����
д��������̼�������ϳɼ״�Һ����Ȼ�ѧ����ʽ ��
��3���״�--����ȼ�ϵ��������ϡ��������������Ϊ�������ʣ�����ϡ�������������ڸ������ܴ���O2-��
�ٸ��������ķ�Ӧ��
����ϡ������������Ĺ��������У�O2-�����ƶ������� ��
��4����֪��Ӧ2CH30H?CH3OCH3��g��+H2O��g��ij�¶��µ�ƽ�ⳣ��Ϊ400���� �¶��£����ܱ������м���CH3OH����Ӧ��ijʱ�̲�ø���ֵ�Ũ�����£�
�ٱȽϴ�ʱ�����淴Ӧ���ʵĴ�Сv�� v�� �����������������=������
��������CH3OH��10min��Ӧ�ﵽƽ�⣬��ʱ���ڷ�Ӧ����v��CH30H��= ��
��1��ͨ��������--��ѭ�����տ��Ƶ�ȼ�ϵ�ص�ȼ��������

�ٸ�ѭ�����չ��̵��ܷ�Ӧ����ʽΪ
���û�ѧƽ���ƶ���ԭ����������HI�ֽⷴӦ��ʹ��Ĥ��Ӧ�������H2��Ŀ����
��2��������̼�ǵ�������ЧӦ��������ף�Ŀǰ���Ǵ���������̼�ķ���֮һ��ʹ����������Ӧ�ϳɼ״�����֪�������״�ȼ�յ��Ȼ�ѧ����ʽ���£�
2H2��g��+O2��g��=2H2O��l������H=-283.0kJ?mol-1����
2CH3OH��1��+3O2��g����2CO2��g��+4H2O��1������H=-726.0kJ?mol-1����
д��������̼�������ϳɼ״�Һ����Ȼ�ѧ����ʽ
��3���״�--����ȼ�ϵ��������ϡ��������������Ϊ�������ʣ�����ϡ�������������ڸ������ܴ���O2-��
�ٸ��������ķ�Ӧ��
����ϡ������������Ĺ��������У�O2-�����ƶ�������
��4����֪��Ӧ2CH30H?CH3OCH3��g��+H2O��g��ij�¶��µ�ƽ�ⳣ��Ϊ400���� �¶��£����ܱ������м���CH3OH����Ӧ��ijʱ�̲�ø���ֵ�Ũ�����£�
| ���� | CH3OH | CH3OCH3 | H2O |
| Ũ��/��mol?L-1�� | 0.44 | 0.6 | 0.6 |
��������CH3OH��10min��Ӧ�ﵽƽ�⣬��ʱ���ڷ�Ӧ����v��CH30H��=

 ��2012?����ģ�⣩��Դ��ȱ���������ٵ��ش����⣮�״���һ�ֿ�������Դ�����й㷺�Ŀ�����Ӧ��ǰ������˼״�����Ϊ21���͵�����ȼ�ϣ�
��2012?����ģ�⣩��Դ��ȱ���������ٵ��ش����⣮�״���һ�ֿ�������Դ�����й㷺�Ŀ�����Ӧ��ǰ������˼״�����Ϊ21���͵�����ȼ�ϣ�