��Ŀ����
�ס�������ʵ��С��ֱ���С�Na2CO3 ��NaCl �������Na2CO3 �����ⶨ����ʵ�飺
(1)����ķ����ǣ�����֪Ũ������μӵ�һ�������Ļ�� ���������������Һ��������ⶨ̼���Ƶĺ������� ʵ������г����ᡢ������ˮ���⣬�жϷ�Ӧ�Ƿ���ȫ�����õ����Լ���___________ ��
(1)����ķ����ǣ�����֪Ũ������μӵ�һ�������Ļ�� ���������������Һ��������ⶨ̼���Ƶĺ������� ʵ������г����ᡢ������ˮ���⣬�жϷ�Ӧ�Ƿ���ȫ�����õ����Լ���___________ ��

(2)����ķ����ǣ�����Ʒ��ϡ���ᷴӦ���ⶨ��Ӧ������ CO2������������㲢�������Ʒ��Na2CO3������������ͼװ�òⶨ������CO2���������������ͼ��װ�����Ͳ��������________��ʹ��Ӧǰ���Ͳ��Ϸ����� ѹǿ��������ѹ��ͬ����������________��
(3)���������һ����ס����������ͬ��ʵ�鷽�����ⶨ������е�Na2CO3��������Ʒ�����____________��
(3)���������һ����ס����������ͬ��ʵ�鷽�����ⶨ������е�Na2CO3��������Ʒ�����____________��
(1)ָʾ��
(2)��ֹ������̼����ˮӰ����������IJⶨ���ɼ� ���ƶ������ܣ�ʹ�Ͳ��������ܵ�Һ�洦��ͬһˮƽ��
(3)ȡһ�����Ļ�����ܽ�����������CaCl2��Ȼ����ˡ�ϴ�ӡ���ɡ�����̼��Ƶ���������̼��Ƶ��������Na2CO3������
(2)��ֹ������̼����ˮӰ����������IJⶨ���ɼ� ���ƶ������ܣ�ʹ�Ͳ��������ܵ�Һ�洦��ͬһˮƽ��
(3)ȡһ�����Ļ�����ܽ�����������CaCl2��Ȼ����ˡ�ϴ�ӡ���ɡ�����̼��Ƶ���������̼��Ƶ��������Na2CO3������

��ϰ��ϵ�д�
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
�����Ŀ
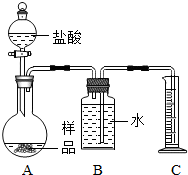 ��2013?�ٳ���ģ�⣩�ס�������ʵ��С��ֱ���С��ⶨNa2CO3��NaCl�������Na2CO3��������ʵ�飮
��2013?�ٳ���ģ�⣩�ס�������ʵ��С��ֱ���С��ⶨNa2CO3��NaCl�������Na2CO3��������ʵ�飮
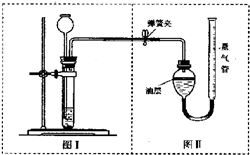 �ס�������ʵ��С��ֱ���С�Na2CO3��NaCl�������Na2CO3�����ⶨ����ʵ�飺
�ס�������ʵ��С��ֱ���С�Na2CO3��NaCl�������Na2CO3�����ⶨ����ʵ�飺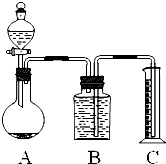 �ס�������ʵ��С��ֱ���С��ⶨNa2CO3��NaCl�������Na2CO3��������ʵ�飮
�ס�������ʵ��С��ֱ���С��ⶨNa2CO3��NaCl�������Na2CO3��������ʵ�飮