��Ŀ����
��1���������Ͷ�������Ļ�������У�������Ԫ�ص���������Ϊ60%����û�������������Ͷ��������������Ϊ______��������ж����������������Ϊ______��
��2��һ��������A��Һ��111g�Ȼ�����Һǡ����ȫ��Ӧ�����ɰ�ɫ����B����ѧ����ʽΪ��A+CaCl2�TB��+2NaCl��
��A�Ļ�ѧʽΪ______��
���˳�����B���ٺ�ɣ�������10g����CaCl2��Һ�����ʵ���������Ϊ���٣�______��
��2��һ��������A��Һ��111g�Ȼ�����Һǡ����ȫ��Ӧ�����ɰ�ɫ����B����ѧ����ʽΪ��A+CaCl2�TB��+2NaCl��
��A�Ļ�ѧʽΪ______��
���˳�����B���ٺ�ɣ�������10g����CaCl2��Һ�����ʵ���������Ϊ���٣�______��
��1���������Ͷ�������Ļ�������У�������Ԫ�ص���������Ϊ60%������Ԫ�ص���������Ϊ��1-60%=40%���������������Ϊ100g������Ԫ�ص�����Ϊ100g��40%=40g���ʶ������������Ϊ��
=80g��������������Ϊ100g-80g=20g���������Ͷ��������������Ϊ��20g��80g=1��4����������ж����������������Ϊ��
��100%=80%�����1��4��80%��
��2����A�����Ȼ��Ʒ�Ӧ���ɰ�ɫ�������Ȼ��ƣ���A��̼���ƣ�B��̼��ƣ����Na2CO3��
�����Ȼ��Ƶ�����Ϊx
Na2CO3+CaCl2�TCaCO3��+2NaCl
111 100
x 10g
=
x=11.1g
��CaCl2��Һ�����ʵ���������Ϊ��
��100%=10%
���10%��
| 40g |
| 50% |
| 80g |
| 100g |
��2����A�����Ȼ��Ʒ�Ӧ���ɰ�ɫ�������Ȼ��ƣ���A��̼���ƣ�B��̼��ƣ����Na2CO3��
�����Ȼ��Ƶ�����Ϊx
Na2CO3+CaCl2�TCaCO3��+2NaCl
111 100
x 10g
| 111 |
| x |
| 100 |
| 10g |
��CaCl2��Һ�����ʵ���������Ϊ��
| 11.1g |
| 111g |
���10%��

��ϰ��ϵ�д�
�����Ŀ
 ij����С���о���Ӱ��H2O2 ����O2���������ء��Ŀ��⣮ͨ���������ϣ�����������²��룮
ij����С���о���Ӱ��H2O2 ����O2���������ء��Ŀ��⣮ͨ���������ϣ�����������²��룮[�������]�����ͷ�Ӧ��Ũ�ȶ���Ӱ��H2O2 ����O2�Ŀ���
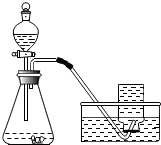
[ʵ�����]ʵ��װ��ͼ��ͼ��ʾ��
ʵ����������װ�����������ã�����Һ©���е�Һ�������ƿ�У������ռ�һƿ�ų������壮
ʵ���¼��
| ʵ���� | �� | �� | �� |
| ��Ӧ�� | 5%H2O2100mL | 5%H2O2 | 3%H2O2100mL |
| ������� | 0.5g�Ȼ��ƹ��� | 0.5g�������� | |
| �ռ������ʱ�� | 165s | 46s | 80s |
��2��ʵ����м���H2O2�����Ϊ
��3��ʵ����м���Ĺ��������Ϊ
[����]��̽�����̵ó��Ľ�����
[��˼]H2O2�ڳ����·ֽ��������MnO2��Ӧ���Լӿ죮С�������Ϊ�˸��õ�֤���������̺��Ȼ����Ƿ��H2O2 ����O2������Ӱ�죬��Ӧ������һ��Ա�ʵ�飮��ʵ��ѡ�õ�ҩƷ������Ϊ
[ʵ����չ]С�����ռ��������������������ʵ�飮
��1�����������ķ�����
��2��С�췢���ø������������������ʵ�飬�������ԣ�������ֽ����ԭ����
 ij��ѧ��ȤС������ʦָ���£������ˡ�H2O2����O2�Ŀ�����ʲô�����йء���ʵ�飮��������ش��������⣮��ʵ�����20�������½��У�
ij��ѧ��ȤС������ʦָ���£������ˡ�H2O2����O2�Ŀ�����ʲô�����йء���ʵ�飮��������ش��������⣮��ʵ�����20�������½��У�