��Ŀ����
��ʦ����þ���ڿ�����ȼ�գ�ϸ�ĵ�ͬѧ���֣�ȼ�պ����������˰�ɫ��ĩ״�������⣬������������ɫ���壮ͬѧ�Dz�����֪������þ��ˮֻ������Ӧ���������ǶԵ��ư�ɫ������������µ�̽������ش����е����⣺
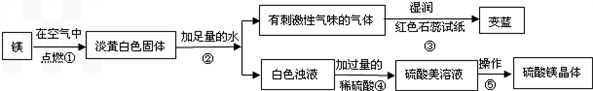
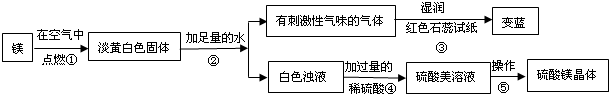
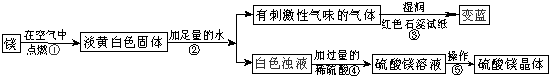
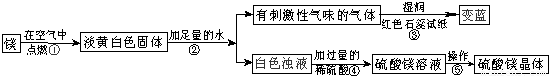
��1��þ�ڿ�����ȼ�յ������У� _________ ���а�ɫ���̡� _________ ���е��ư�ɫ�������ɣ�þ��������Ӧ�Ļ�ѧ����ʽΪ�� ___ ______ ��
��2���ɲ����۵�����˵���������� _________ ���ѧʽ���������������˽������������þȼ�ղ����еĵ���ɫ����Mg3N2��ˮ��Ӧ����������þʱ���ɵģ��仯ѧ����ʽΪ�� ___ ______ ��
��3�������ݰ����ľ�����������ǣ�����Ũ�������¡� _________ ��
��2���ɲ����۵�����˵���������� _________ ���ѧʽ���������������˽������������þȼ�ղ����еĵ���ɫ����Mg3N2��ˮ��Ӧ����������þʱ���ɵģ��仯ѧ����ʽΪ�� ___ ______ ��
��3�������ݰ����ľ�����������ǣ�����Ũ�������¡� _________ ��
��1������ҫ�۵İ⣻ �ų��������ȣ� 2Mg+O2 2MgO��
2MgO��
��2��NH3�� Mg3N2+6H2O=3Mg(OH)2+2NH3����
��3���ᾧ��
 2MgO��
2MgO����2��NH3�� Mg3N2+6H2O=3Mg(OH)2+2NH3����
��3���ᾧ��

��ϰ��ϵ�д�
 ���Ž�������С״Ԫϵ�д�
���Ž�������С״Ԫϵ�д�
�����Ŀ