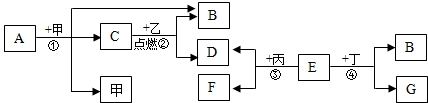
��Ŀ����
ij��ѧ��ȤС��Ϊ�˲ⶨһ��ʯ��ʯ��Ʒ��̼��Ƶ�����������ȡ��8gʯ��ʯ��Ʒ����40gϡ�����4�μ�����Ʒ�У����ʼȲ������ᷴӦ��Ҳ������ˮ������ַ�Ӧ���ˡ�����Ȳ���������������ʵ���������ң�����ѧ����ʽΪ��CaCO3+2HCl�TCaCl2+H2O+CO2����
��1���ϱ���M����ֵӦΪ
��2����Ʒ��̼��Ƶ���������Ϊ
��3������������100t����ʯ��ʯ��Ӧ���ܵõ�������̼�������Ƕ��٣�
| ϡ��������� | ʣ���������� |
| ��һ�μ���10g | 5.5g |
| �ڶ��μ���10g | M |
| �����μӰ�10g | 1.2g |
| ���ĴμӰ�10g | 1.2g |
��2����Ʒ��̼��Ƶ���������Ϊ
��3������������100t����ʯ��ʯ��Ӧ���ܵõ�������̼�������Ƕ��٣�
���㣺���ݻ�ѧ��Ӧ����ʽ�ļ���
ר�⣺�ۺϼ��㣨ͼ���͡������͡��龰�ͼ����⣩
��������һ�μ���10gϡ�����ʣ������������5.5g����Ӧ��̼�������Ϊ��8g-5.5g=2.5g��˵��10gϡ�����ܺ�2.5g̼���ǡ����ȫ��Ӧ��
�ڶ��μ���10gϡ��������̼���������ʣ����������Ӧ��Ϊ��5.5g-2.5g=3g��
�����μӰ�10gϡ�����ʣ������������1.2g��˵��������ʵ���ʯ��ʯ��Ʒ�е�̼�������ȫ��Ӧ��
�ٸ��ݱ����ṩ�����ݿ��Խ�����ط���ļ��㣮
�ڶ��μ���10gϡ��������̼���������ʣ����������Ӧ��Ϊ��5.5g-2.5g=3g��
�����μӰ�10gϡ�����ʣ������������1.2g��˵��������ʵ���ʯ��ʯ��Ʒ�е�̼�������ȫ��Ӧ��
�ٸ��ݱ����ṩ�����ݿ��Խ�����ط���ļ��㣮
����⣺��1�����������ݿ�֪�����Ĵ�ʵ���У��ڶ��μ���10gϡ��������̼���������ʣ����������Ӧ��Ϊ��5.5g-2.5g=3g��������M����ֵӦΪ3g�����3g��
��2��ʯ��ʯ��Ʒ��̼��Ƶ�����Ϊ��8g-1.2g=6.8g��
ʯ��ʯ��Ʒ��̼��Ƶ���������Ϊ��
��100%=85%
��ʯ��ʯ��Ʒ��̼��Ƶ���������Ϊ85%��
�ʴ�Ϊ��85%��
��3�����ܵõ�������̼������Ϊx��
CaCO3+2HCl�TCaCl2+H2O+CO2��
100 44
100t��85% x
=
x=37.4t
���ܵõ�������̼������Ϊ37.4t��
��2��ʯ��ʯ��Ʒ��̼��Ƶ�����Ϊ��8g-1.2g=6.8g��
ʯ��ʯ��Ʒ��̼��Ƶ���������Ϊ��
| 6.8g |
| 8g |
��ʯ��ʯ��Ʒ��̼��Ƶ���������Ϊ85%��
�ʴ�Ϊ��85%��
��3�����ܵõ�������̼������Ϊx��
CaCO3+2HCl�TCaCl2+H2O+CO2��
100 44
100t��85% x
| 100 |
| 100t��85% |
| 44 |
| x |
x=37.4t
���ܵõ�������̼������Ϊ37.4t��
������������Ҫ����ѧ�����ü��跨�ͻ�ѧ����ʽ���м�����ƶϵ��������Ƚϼ�Ҫע�����Ĺ淶�Ժ�ȷ�ԣ�

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
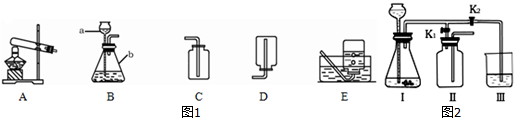
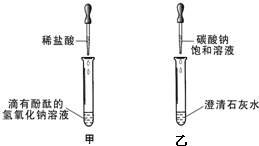 ͬѧ����ѧϰ��Ļ�ѧ����ʱ��������ͼ��ʾ�ļ�������ʵ�飮
ͬѧ����ѧϰ��Ļ�ѧ����ʱ��������ͼ��ʾ�ļ�������ʵ�飮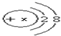 ����X����11���������ʾ��
����X����11���������ʾ��