��Ŀ����
�ҹ���ѧ�Һ�°����ġ������Ƽ���������Ժ�ˮ��ɹ�Ρ��õ����Ȼ���Ϊԭ�ϣ�ͬʱ�Ƶ�NaHCO3��NH4Cl��Ʒ����̼�������Լ��ȼ��ֽ�õ������ͼ1����
��1��ͨ����Ӧ�ڿɵõ�̼�����ƣ���ѧ����ʽΪ��NaCl+NH4HCO3=NaHCO3+NH4Cl���÷�Ӧ���� ��Ӧ���ͣ�
��2��NaCl��NaHCO3��NH4Cl���ܽ��������ͼ2��
�ش��������⣺
��P��ĺ��壺 ��
��̼�������ܽ�������ڴ���60��͡���ʧ���ˣ���˵��ԭ�� ��
��20��ʱ����100gˮ�м���11.7gNaCl��15.8gNH4HCO3������Ӧ���Ƶ�NaHCO3�������ϴ���Һ���������������Ϊ g��
���𰸡���������1�����ݷ�Ӧ�����������ص������Ӧ���ͣ�
��2���ٸ����ܽ�����߽����ʾ���¶������ʵ��ܽ����ȷ�����
�ڸ�����Ϣ��̼�������Լ��ȼ��ֽ�õ����������
�۸���20��ʱ NaCl��NH4HCO3���ܽ�ȿ�֪��������ȫ�ܽ⣬�ɷ���ʽ������֮���������ϵ��֪NaCl��NH4HCO3ǡ����ȫ��Ӧ��������NaCl�������������ɵ�NaHCO3��NH4Cl����������ϸ��¶������ʵ��ܽ�ȷ���������������ʼ�������
����⣺��1���÷�Ӧ�����ֻ�����������ɷ֣����������µĻ�����ķ�Ӧ���Ǹ��ֽⷴӦ��
��2����P��ĺ����ǣ�20��ʱNH4Cl��NaCl���ܽ�ȶ���36g��
����Ϊ��̼�������Լ��ȼ��ֽ�õ�������̼�������ܽ�������ڴ���60��͡���ʧ���ˣ�
�۽⣺��1�����ݻ�ѧ����ʽ��������ϵ����֪����NaCl��NH4HCO3ǡ����ȫ��Ӧ�������ɵ�NaHCO3������Ϊx��NH4Cl������Ϊy
NaCl+NH4HCO3�TNaHCO3+NH4Cl
58.5 79 84
11.7g x
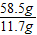 =
=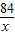 x=16.8g
x=16.8g
�������е��ܽ�ȱ�����֪����20��ʱ100gˮ���ܹ��ܽ�̼�����Ƶ�����Ϊ9.66g�����������ϴ���Һ��������NaHCO3���������Ϊ16.8g-9.66g=7.14g��
�ʴ�Ϊ����1�����ֽ⣻��2��20��ʱNH4Cl��NaCl���ܽ�ȶ���36g����3��̼�����Ƽ����ֽ�õ����
��4��7.14g��
���������⿼����ѧ�����������������龰��ͼ����Ϣ�������ѧ�ܽ�ȡ�����ʽ��������֪ʶ�ͼ��ܣ�ϸ�ĵ�̽����������������ĿҪ��������������������������
��2���ٸ����ܽ�����߽����ʾ���¶������ʵ��ܽ����ȷ�����
�ڸ�����Ϣ��̼�������Լ��ȼ��ֽ�õ����������
�۸���20��ʱ NaCl��NH4HCO3���ܽ�ȿ�֪��������ȫ�ܽ⣬�ɷ���ʽ������֮���������ϵ��֪NaCl��NH4HCO3ǡ����ȫ��Ӧ��������NaCl�������������ɵ�NaHCO3��NH4Cl����������ϸ��¶������ʵ��ܽ�ȷ���������������ʼ�������
����⣺��1���÷�Ӧ�����ֻ�����������ɷ֣����������µĻ�����ķ�Ӧ���Ǹ��ֽⷴӦ��
��2����P��ĺ����ǣ�20��ʱNH4Cl��NaCl���ܽ�ȶ���36g��
����Ϊ��̼�������Լ��ȼ��ֽ�õ�������̼�������ܽ�������ڴ���60��͡���ʧ���ˣ�
�۽⣺��1�����ݻ�ѧ����ʽ��������ϵ����֪����NaCl��NH4HCO3ǡ����ȫ��Ӧ�������ɵ�NaHCO3������Ϊx��NH4Cl������Ϊy
NaCl+NH4HCO3�TNaHCO3+NH4Cl
58.5 79 84
11.7g x
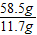 =
=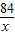 x=16.8g
x=16.8g �������е��ܽ�ȱ�����֪����20��ʱ100gˮ���ܹ��ܽ�̼�����Ƶ�����Ϊ9.66g�����������ϴ���Һ��������NaHCO3���������Ϊ16.8g-9.66g=7.14g��
�ʴ�Ϊ����1�����ֽ⣻��2��20��ʱNH4Cl��NaCl���ܽ�ȶ���36g����3��̼�����Ƽ����ֽ�õ����
��4��7.14g��
���������⿼����ѧ�����������������龰��ͼ����Ϣ�������ѧ�ܽ�ȡ�����ʽ��������֪ʶ�ͼ��ܣ�ϸ�ĵ�̽����������������ĿҪ��������������������������

��ϰ��ϵ�д�
�����Ŀ


