��Ŀ����
����![]() ������ܽ�ȱ����ܽ�����ش��������⣺
������ܽ�ȱ����ܽ�����ش��������⣺
|
| 0 | 20 | 40 | 60 | 80 | ||||||||||||||
| KNO3 | 13.3 | 31.6 | 63.9 | 110 | 169 | ||||||||||||||
| NaCl | 35.7 | 36.0 | 36.6 | 37.3 | 38.4 | ||||||||||||||
| Ca(OH)2 | 0.173 | 0.165 | 0.121 | 0.116 | 0.094 |
��1��B��______���ܽ�����ߡ�
��2��40��ʱ���Ȼ��Ƶ��ܽ��______������ڡ�����С�ڡ����ڡ�������ص��ܽ�ȡ�
��3����ʹ����صIJ�������Һת��Ϊ������Һ�����Բ�ȡ�ķ���֮һ�� ��
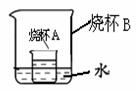
��4������ͼ��ʾ���ձ�A���DZ��͵�����������Һ�����ձ�B�м�����ʯ�Һ��ձ�A�б���ǣ����ܵ�ԭ����______������ţ���
A����Ӧ����ˮ��������������
B����Ӧ���ȣ��¶����ߣ����������ܽ�Ƚ���
C����ʯ����ˮ��Ӧ���ɵ��������Ʋ����ܽ�
 |
��1��NaCl ��2��С�� ��3�����»��������� ��4��B

��ϰ��ϵ�д�
�����Ŀ
 ��2011?��Զ������������ܽ�ȱ����ܽ�����ش��������⣺
��2011?��Զ������������ܽ�ȱ����ܽ�����ش��������⣺ A����Ӧ����ˮ��������������
A����Ӧ����ˮ�������������� ��2013?ͨ����һģ��ˮ����Һ�������������������ʮ����Ҫ�����ã�����������ܽ�ȱ����ܽ�����ߣ�ͼ1�����ش��������⣺
��2013?ͨ����һģ��ˮ����Һ�������������������ʮ����Ҫ�����ã�����������ܽ�ȱ����ܽ�����ߣ�ͼ1�����ش��������⣺ ��2013?��ɽ����ģ������������벻����Һ������������ܽ�ȱ����ܽ�����ߣ��ش��������⣺
��2013?��ɽ����ģ������������벻����Һ������������ܽ�ȱ����ܽ�����ߣ��ش��������⣺ ˮ����Һ�������������������ʮ����Ҫ�����ã�����������ܽ�ȱ���
ˮ����Һ�������������������ʮ����Ҫ�����ã�����������ܽ�ȱ���

