��Ŀ����
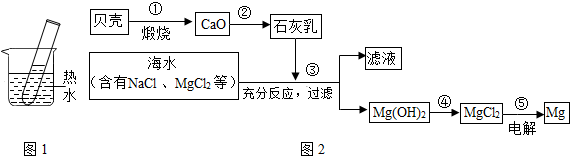
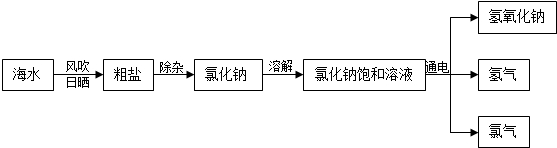
������һ�������Դ���⣬���ǿ��ԴӺ�ˮ����ȡ�Ȼ��ƣ�ijͬѧ���������̴Ӻ�����ɳ���Ȼ��Ƶ�ʳ��ˮ����ȡ20g���������Ȼ��ƵĴ��Σ����ⶨ�ô������Ȼ��Ƶ�������������Ҫ�������̣�����Ҫ��Ӧ��CaCl2+Na2CO3��CaCO3��+2NaCl��
��1�������ǽ��������з��룬������������������������______��
��2��20g�������Ȼ��Ƶ����ʵ���Ϊ______��
��3���ô������Ȼ��Ƶ���������Ϊ______��
���𰸡���������1�������Ƿ��벻���Թ����Һ���һ�ֳ��õķ�����
��2���������ɳ�����������ϻ�ѧ����ʽ����������Ȼ��Ƶ�����������������Ȼ��Ƶ����ʵ�����
��3�����ݣ�2���м���������������Ȼ��Ƶ�������������ʵ����������ļ��㹫ʽ��������Ȼ��Ƶ�����������
����⣺��1�������Ƿ��벻���Թ����Һ���һ�ֳ��õķ����������Ա��������ʱ��벻������Һ�壻
��2������ͼ����ʾ����֪��������5g̼��ƣ�����̼��Ƶ����ʵ���Ϊ��5g÷100g/mol=0.05mol���ʣ�
��20g�������Ȼ��Ƶ����ʵ���Ϊx����
CaCl2+Na2CO3��CaCO3��+2NaCl
1 1
x 0.05mol

��ã�x=0.05mol��
��3���ô������Ȼ��Ƶ���������Ϊ�� ×100%=72.25%��
×100%=72.25%��
�𣺣�2��20g�������Ȼ��Ƶ����ʵ���Ϊ0.05mol����3���ô������Ȼ��Ƶ���������Ϊ72.25%��
������������Ҫ���麬�������ʵĻ�ѧ����ʽ������������������ļ��㣬�ѶȽϴ�
��2���������ɳ�����������ϻ�ѧ����ʽ����������Ȼ��Ƶ�����������������Ȼ��Ƶ����ʵ�����
��3�����ݣ�2���м���������������Ȼ��Ƶ�������������ʵ����������ļ��㹫ʽ��������Ȼ��Ƶ�����������
����⣺��1�������Ƿ��벻���Թ����Һ���һ�ֳ��õķ����������Ա��������ʱ��벻������Һ�壻
��2������ͼ����ʾ����֪��������5g̼��ƣ�����̼��Ƶ����ʵ���Ϊ��5g÷100g/mol=0.05mol���ʣ�
��20g�������Ȼ��Ƶ����ʵ���Ϊx����
CaCl2+Na2CO3��CaCO3��+2NaCl
1 1
x 0.05mol

��ã�x=0.05mol��
��3���ô������Ȼ��Ƶ���������Ϊ��
 ×100%=72.25%��
×100%=72.25%���𣺣�2��20g�������Ȼ��Ƶ����ʵ���Ϊ0.05mol����3���ô������Ȼ��Ƶ���������Ϊ72.25%��
������������Ҫ���麬�������ʵĻ�ѧ����ʽ������������������ļ��㣬�ѶȽϴ�

��ϰ��ϵ�д�
 ����ѵ�����⿼ϵ�д�
����ѵ�����⿼ϵ�д�
�����Ŀ