��Ŀ����
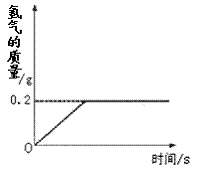
�⻯ﮣ�LiH��Ϊ����״��ɫ�����壬��������������������Դ��ij��ѧ��ȤС��ȡһ���������⻯���99.4��ˮ��Ӧ����������������뷴Ӧʱ��Ĺ�ϵ��ͼ��ʾ����ش��������⣺
|
LiH��H2O= ��H2�� ��
��X�Ļ�ѧʽΪ ��
��.�⻯���Li��HԪ��������Ϊ ��
��3����Ӧ��������������������Ϊ_______g��
��4��ȡ���⻯﮵�����Ϊ���٣�
��LiOH��1�֣��� 7:1��2�֣�
��0.2��2�֣�(4)��LiH������Ϊx��������1�֣�
LiH��H2O=LiOH��H2��
8 2
x 0.2g������2�֣�
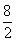 =
=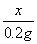 ������1�֣�
������1�֣�
��֮�ã�x=0.8g������1�֣�
�� ȡ��LiH������Ϊ0.8g������ʹ�ȫ�Ը�1�֣����д�����֡���

��ϰ��ϵ�д�
�����Ŀ
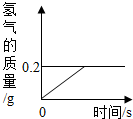 ��2013?�����ģ���⻯ﮣ�LiH��Ϊ����״��ɫ�����壬��������������������Դ��ij��ѧ��ȤС��ȡһ���������⻯���ˮ��Ӧ���õ�100g������ﮣ�LiOH����Һ��ͬʱ������������LiH+H2O=LiOH+H2���������������������뷴Ӧʱ��Ĺ�ϵ��ͼ��ʾ�����������������Һ����������Ϊ���٣�
��2013?�����ģ���⻯ﮣ�LiH��Ϊ����״��ɫ�����壬��������������������Դ��ij��ѧ��ȤС��ȡһ���������⻯���ˮ��Ӧ���õ�100g������ﮣ�LiOH����Һ��ͬʱ������������LiH+H2O=LiOH+H2���������������������뷴Ӧʱ��Ĺ�ϵ��ͼ��ʾ�����������������Һ����������Ϊ���٣�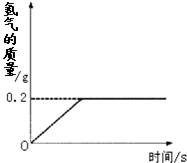 �⻯ﮣ�LiH��Ϊ����״��ɫ�����壬��������������������Դ��ij��ѧ��ȤС��ȡһ���������⻯���99.4��ˮ��Ӧ����������������뷴Ӧʱ��Ĺ�ϵ��ͼ��ʾ����ش��������⣺
�⻯ﮣ�LiH��Ϊ����״��ɫ�����壬��������������������Դ��ij��ѧ��ȤС��ȡһ���������⻯���99.4��ˮ��Ӧ����������������뷴Ӧʱ��Ĺ�ϵ��ͼ��ʾ����ش��������⣺ ���� LiOH + H2��������80 g�⻯���������ˮ��Ӧ����������������
���� LiOH + H2��������80 g�⻯���������ˮ��Ӧ����������������