��Ŀ����
ij��ѧ��ȤС���ͬѧ��������װ�ý���ʵ������ȡ�����̽����
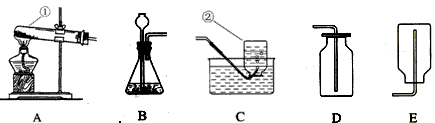
��1��д��ָ�����������ƣ���________ ����________ ��
��2��Сǿ�ù���������Һ�Ͷ���������ȡ���ռ���������Ӧѡ�õ�װ����________������ţ���ͬ����д���÷�Ӧ�Ļ�ѧ����ʽ_________________��ʵ���������ø������Ҳ������ȡ�����壬��Ӧѡ��ķ���װ����_________��д���÷�Ӧ�Ļ�ѧ����ʽ_________________��
��3��С���ռ�������̼ѡ��Dװ�ã�����ѡ��C��Eװ�ã��ԴӶ�����̼���ʵĽǶȷ�����ԭ����_____________��д��ʵ������ȡ������̼�Ļ�ѧ����ʽ________________��
��4��Сʯ�������ͼװ����ȡ������̼���壬���ж�����ϰ��������_________�����ø�װ�û�������ȡ��������_________________��
��2��Сǿ�ù���������Һ�Ͷ���������ȡ���ռ���������Ӧѡ�õ�װ����________������ţ���ͬ����д���÷�Ӧ�Ļ�ѧ����ʽ_________________��ʵ���������ø������Ҳ������ȡ�����壬��Ӧѡ��ķ���װ����_________��д���÷�Ӧ�Ļ�ѧ����ʽ_________________��
��3��С���ռ�������̼ѡ��Dװ�ã�����ѡ��C��Eװ�ã��ԴӶ�����̼���ʵĽǶȷ�����ԭ����_____________��д��ʵ������ȡ������̼�Ļ�ѧ����ʽ________________��
��4��Сʯ�������ͼװ����ȡ������̼���壬���ж�����ϰ��������_________�����ø�װ�û�������ȡ��������_________________��
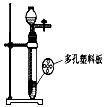
(1)�Թܣ�����ƿ��
(2)BD����BC����2H2O2 2H2O + O2���� A��2KMnO4
2H2O + O2���� A��2KMnO4 K2MnO4 + MnO2 + O2��
K2MnO4 + MnO2 + O2��
(3) CO2���ܶȱȿ�������������ˮ��CaCO3 + 2HCl === CaCl2 + H2O + CO2��
(4)����ʯ��ʯ�������ʯ����H2
(2)BD����BC����2H2O2
 2H2O + O2���� A��2KMnO4
2H2O + O2���� A��2KMnO4 K2MnO4 + MnO2 + O2��
K2MnO4 + MnO2 + O2�� (3) CO2���ܶȱȿ�������������ˮ��CaCO3 + 2HCl === CaCl2 + H2O + CO2��
(4)����ʯ��ʯ�������ʯ����H2

��ϰ��ϵ�д�
�����Ŀ
 12��̼�����ƣ�NaHCO3���׳�С�մ�����ʳƷ��ҽҩ��ҵ��ij��ѧ��ȤС���ͬѧ��̼�����Ƶ����ʽ���̽����
12��̼�����ƣ�NaHCO3���׳�С�մ�����ʳƷ��ҽҩ��ҵ��ij��ѧ��ȤС���ͬѧ��̼�����Ƶ����ʽ���̽����


 ��2012?��̨��ij��ѧ��ȤС���ͬѧ����ͼ��ʾװ�ý���ʵ�飨װ�����������ã����ȹر�ֹˮ�У�������������������Һ������ƿ�У�������յ�������̼���ٴ�ֹˮ�У�
��2012?��̨��ij��ѧ��ȤС���ͬѧ����ͼ��ʾװ�ý���ʵ�飨װ�����������ã����ȹر�ֹˮ�У�������������������Һ������ƿ�У�������յ�������̼���ٴ�ֹˮ�У� ij��ѧ��ȤС���ͬѧ��ʵ����������������Ϊ8%������������Һ��������ⶨijϡ���������ʵ�����������
ij��ѧ��ȤС���ͬѧ��ʵ����������������Ϊ8%������������Һ��������ⶨijϡ���������ʵ�����������