��Ŀ����
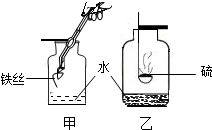
��8�֣�ˮ������֮Դ������֮���������౦�����Ȼ��Դ������ÿ���˶�Ҫ�����ͽ�Լ����1�����о�ˮ�����У�ͨ�����ڳ�ȥˮ�����������ʵ��� ��������ĸ����ͬ����ˮ�̶���ߵ��� ��
A������ B����� C������ D����
��2������ˮ�����ö������ȣ�ClO2��������������Ԫ�صĻ��ϼ��� ��
��3��Ӳˮ��������������������㡣
������Ӳˮ����ˮ�����õ�������______�� ����������ν���ˮ��Ӳ�� ��
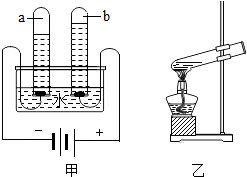
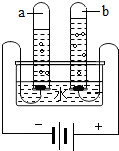
��4�����ʵ���У������븺���������Ϊ ������������μ��� ����ⷴӦ�Ļ�ѧ����ʽ�� ��
��1��A ��C
(2) +4
(3) �ٷ���ˮ����п�����ˮ���Ķ��� �����
��4��1:2 ���������ľ������ȼ����������2H2O 2 H2�� + O2��������:
2 H2�� + O2��������:
��
(2) +4
(3) �ٷ���ˮ����п�����ˮ���Ķ��� �����
��4��1:2 ���������ľ������ȼ����������2H2O
 2 H2�� + O2��������:
2 H2�� + O2��������:��

��ϰ��ϵ�д�
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
�����Ŀ
 ˮ������֮Դ������֮���������౦�����Ȼ��Դ������ÿ���˶�Ҫ����ˮ������ˮ����Լ��ˮ��
ˮ������֮Դ������֮���������౦�����Ȼ��Դ������ÿ���˶�Ҫ����ˮ������ˮ����Լ��ˮ��
