��Ŀ����
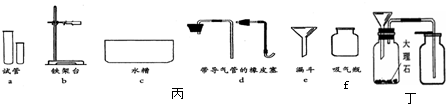
ij��ѧ��ȤС��������ͼװ��̽���������ȡԭ�������������ʣ����װ��ͼ���ش��������⣺
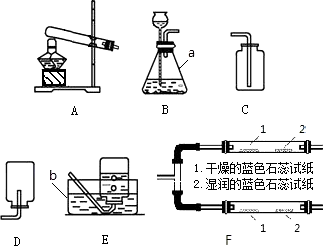
(1)д����ͼ������a�����ƣ�________��ʵ������ȡ������Ӧѡ���װ����________(����ͼ��ĸ)��
(2)��װ��A��ȡ�����Ļ�ѧ����ʽΪ________����Eװ���ռ�����������ԭ�������________(дһ��)��
(3)С����װ��F̽��CO2��ijЩ���ʣ�����������CO2ͨ��װ��F�۲쵽���������沣������ʪ���ʯ����ֽ��죬���沣���������������ݴ�˵��������̼�������У���________����________��
�𰸣���1����ƿ B
��2��2KCIO3 = MnO2��= 2KCI + 3O2�� ��δ���Թܼ������п����ž������ܿڸճ������ݾͿ�ʼ�ռ��������ڼ���ƿ��ˮû��װ�����ռ��������������ƿ���ǵ��ţ�����Ƭû�и���ƿ��
��3��������̼���������壻������ˮ

��ϰ��ϵ�д�
 �ִʾ�ƪ��ͬ�����Ĵ��ϵ�д�
�ִʾ�ƪ��ͬ�����Ĵ��ϵ�д�
�����Ŀ



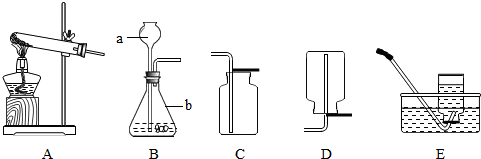
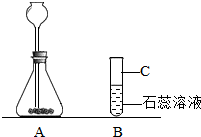
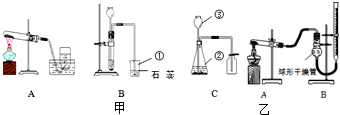
 ��2007?���ij��ѧ��ȤС��������ͼװ�ý��б���NaCl��Һ�ĵ��ʵ�飬���Բ��ֲ������̽����
��2007?���ij��ѧ��ȤС��������ͼװ�ý��б���NaCl��Һ�ĵ��ʵ�飬���Բ��ֲ������̽����