��Ŀ����
19������HCl��CaCl2�Ļ����Һ��Ϊ�˷��������Һ��HCl��CaCl2�������������������ͼ1��ʾʵ�鷽������ʵ�����ݡ�ʵ�鹲��¼���������ݣ�
�ڢ��飬����������ȫ��NaOH��Һ��������4.4g��
�ڢ��飬������ȫ�����ˡ�ϴ�ӡ���ɺ�õ����������Ϊ10g��
����ʵ����Ƽ��й����ݽ��з�������㣺
��1���������������Ϊ4.4g����ֻд�����
��2�������Һ��CaCl2����������Ϊ���٣���д��������̣�
��3������100g�����Һ�в��ϼ���10.6% ��Na2CO3����Һ�����㻭������Na2CO3��Һ����������������������Ĺ�ϵͼ��ͼ2����

���� ��1�����������֪�����ɶ�����̼������Ϊ4.4g�����Ծݴ˽��̼���ƺ����ᷴӦ�Ļ�ѧ����ʽ������Ȼ����������
��2������ͼ�����ݿ���֪�����ɳ���������Ϊ10g�����Ծݴ˽��̼���ƺ��Ȼ��Ʒ�Ӧ�Ļ�ѧ����ʽ������Ȼ��Ƶ�������Ȼ������������������ɣ�
��3�����������������ʵ����ʿ���֪������������ڵ������̼��Ʋ������Գ�������ʽ���������Ծݴ˽�������������������ͼ��
��� �⣺��1������������Һ���ӵ�������Ϊ̼���ƺ����ᷴӦ���ɶ�����̼����������Ϊ4.4g��
������Һ�к����Ȼ��������Ϊx�������Ȼ��Ƶ�����Ϊy������̼���Ƶ�����Ϊz��
Na2CO3+2HCl�T2NaCl+H2O+CO2��
106 73 117 44
z x y 4.4g
$\frac{106}{z}$=$\frac{73}{x}$=$\frac{117}{y}$=$\frac{44}{4.4g}$
��ã�x=7.3g
��ã�y=11.7g
��ã�z=10.6g
��2��������Һ��CaCl2������Ϊm�������Ȼ��Ƶ�����Ϊn������̼���Ƶ�����Ϊp��
CaCl2+Na2CO3�TCaCO3��+2NaCl
111 106 100 117
m p 10g n
$\frac{111}{m}$=$\frac{106}{p}$=$\frac{100}{10g}$=$\frac{117}{n}$
��ã�m=11.1g
��ã�n=11.7g
��ã�p=10.6g��
�����Һ��CaCl2����������Ϊ��$\frac{11.1g}{100g}$��100%=11.1%��
�𣺻����Һ��CaCl2����������Ϊ11.1%��
��3�����������������ʵ����ʿ���֪������������ڵ������̼��Ʋ������Գ�������ʽ���������Լ����̼����Ҫ�Ⱥ����ᷴӦ��
��������̼������Һ������Ϊ��10.6g��10.6%=100g��
�Ȼ�������̼������Һ������Ϊ��10.6g��10.6%=100g
����֪������������̼������Һ������Ϊ100g����������100g̼������Һ��Ż�����������������ɳ���������Ϊ10g����ʱһ������̼������Һ������Ϊ100g+100g=200g�����Ծݴ�������ߵó�����Na2CO3��Һ����������������������Ĺ�ϵͼ�����𰸣���
�ʴ�Ϊ����1��7.3g��
��2��11.1%
��3��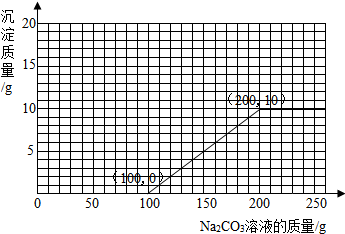
���� Ҫ�����������Ŀ�����ȣ�Ҫ�������Ǹ��ݻ�ѧ��Ӧ����ʽ�ļ��㲽���ʽ���Լ���֮��ص�֪ʶ�ȣ�Ȼ���������������龰�������ѧ�����֪ʶ�ͼ��ܣ�ϸ�µط������Ⲣϸ�ĵ�̽��������������ĿҪ���������ؽ�ɣ�

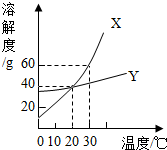 ��ͼ��X��Y���ֹ������ʣ��ᾧˮ�����������ܽ�Ȳ����ţ����ܽ�����ߣ�����˵����ȷ���ǣ�������
��ͼ��X��Y���ֹ������ʣ��ᾧˮ�����������ܽ�Ȳ����ţ����ܽ�����ߣ�����˵����ȷ���ǣ�������| A�� | ��20��ʱ��X��Y��������Һ�����ʵ�����������ͬ | |
| B�� | ���ж�X��Y���ֹ������ʵ��ܽ��� | |
| C�� | ��X�к�������Yʱ��������ȴ�ᾧ�ķ����ᴿY | |
| D�� | ����30���X��Y���߱��͵Ļ����Һ��10��ʱ������X��Y������ |
| A�� | ľ̿��������ȼ�պ����ɺ�ɫ���� | |
| B�� | ���ڿ�����ȼ�ղ�����������ɫ���棬����һ����ɫ��ζ������ | |
| C�� | ������̼���岻����ʹ����ʯ����ֽ���ɫ | |
| D�� | ��˿�ڿ����п��Ծ���ȼ�գ��������䣬������ɫ��Fe2O3���� |
| A�� | ���ˮ�õ�H2��O2����֪ˮ������ԭ�Ӻ���ԭ����ɵ� | |
| B�� | ˮ������������Ԫ��һ�������Ի�ѧ����Ҳһ�� | |
| C�� | Һ̬ˮ���ˮ���������Ӽ������� | |
| D�� | KMnO4��NaCl�����ͼ���һ����ˮ�ж����γ���Һ |

 ѧУ��ѧС���ͬѧΪ�˲ⶨijʯ��ʯ��̼��Ƶ�����������ȡһ��������ʯ��ʯ�����ձ��У���120gϡ��������ɴμ����ձ��У�ʯ��ʯ�е����ʲ������ᷴӦ��Ҳ����ˮ��Ӧ������Ӧ���̵�������ϵ��ͼ��ʾ��x��ʾ����ϡ���������/g��y��ʾ�ձ����������ʵ�������/g������ش��������⣺
ѧУ��ѧС���ͬѧΪ�˲ⶨijʯ��ʯ��̼��Ƶ�����������ȡһ��������ʯ��ʯ�����ձ��У���120gϡ��������ɴμ����ձ��У�ʯ��ʯ�е����ʲ������ᷴӦ��Ҳ����ˮ��Ӧ������Ӧ���̵�������ϵ��ͼ��ʾ��x��ʾ����ϡ���������/g��y��ʾ�ձ����������ʵ�������/g������ش��������⣺