��Ŀ����
ij��ȤС�����������������ĺ��������˲ⶨ������������г�Fe��Fe2O3�⣬������̼���衢�̵�Ԫ�أ���Fe��Fe2O3��������ʶ�����ϡ���ᷴӦ������ȤС���ͬѧ��ȡ40 g������Ƭ�������������У���������39��2%��ϡ���ᣬֱ��ǡ�÷�Ӧ��ȫֹͣ����ϡ���ᣨ�������������������ⷢ����Ӧ����ʵ��������ͼ��ʾ��
��1��������Ƭ�������ʵ���������Ϊ ��
��2��������Һ���������ʵĻ�ѧʽΪ ���������Һ��������������Ϊ g����д��������̣���
��1��70% ��2��FeSO4 ��Fe2(SO4)3 10
��������
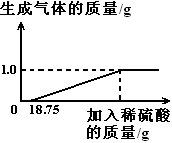
�����������1���۲�ͼʾ����֪ϡ����������Ӧ��������������Ϊ1 g�������������ᷴӦ�ķ���ʽ����������������������������ټ���������ʵ�����������
�����������������Ϊx ��
Fe �� H2SO4 = FeSO4 �� H2��
56 2
x 1 g
56/2 = x/1g�����x = 28g �������������ʵ���������Ϊ28g/40g��100% = 70%
��2����������ֻ��Fe��Fe2O3����ϡ���ᷴӦ������ϡ����ǡ����ȫ��Ӧ��������Һ�е�������ϡ������Fe��Ӧ������FeSO4�Լ�ϡ������Fe2O3��Ӧ������Fe2(SO4)3��
�����⼰ͼ���Կ�����Fe2O3��Ӧ�Ĵ����������Ϊ18��75g��39��2% = 7��35g���跴Ӧ����������������Ϊy
Fe2O3 + 3H2SO4 = Fe2(SO4)3 + 3H2O
294 400
7��35 g y
294/400 =7��35g/y �����y = 10g
����Һ��������������Ϊ10g
���㣺���ݻ�ѧ��Ӧ����ʽ�ļ���

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д� ��2012?��Ӫ��ij��ȤС�����������������ĺ��������˲ⶨ������������г�Fe��Fe2O3�⣬������̼���衢�̵�Ԫ�أ���Fe��Fe2O3��������ʶ�����ϡ���ᷴӦ������ȤС���ͬѧ��ȡ33g������Ƭ�������������У���������39.2%��ϡ���ᣬֱ����Ӧ��ȫ���������������������ⷢ����Ӧ����ʵ��������ͼ��ʾ��
��2012?��Ӫ��ij��ȤС�����������������ĺ��������˲ⶨ������������г�Fe��Fe2O3�⣬������̼���衢�̵�Ԫ�أ���Fe��Fe2O3��������ʶ�����ϡ���ᷴӦ������ȤС���ͬѧ��ȡ33g������Ƭ�������������У���������39.2%��ϡ���ᣬֱ����Ӧ��ȫ���������������������ⷢ����Ӧ����ʵ��������ͼ��ʾ�� ij��ȤС�����������������ĺ��������˲ⶨ������������г�Fe��Fe2O3�⣬������̼���衢�̵�Ԫ�أ���Fe��Fe2O3��������ʶ�����ϡ���ᷴӦ������ȤС���ͬѧ��ȡ33g������Ƭ�������������У���������39.2%��ϡ���ᣬֱ����Ӧ��ȫ���������������������ⷢ����Ӧ����ʵ��������ͼ��ʾ��
ij��ȤС�����������������ĺ��������˲ⶨ������������г�Fe��Fe2O3�⣬������̼���衢�̵�Ԫ�أ���Fe��Fe2O3��������ʶ�����ϡ���ᷴӦ������ȤС���ͬѧ��ȡ33g������Ƭ�������������У���������39.2%��ϡ���ᣬֱ����Ӧ��ȫ���������������������ⷢ����Ӧ����ʵ��������ͼ��ʾ��

